Bisphenols Defined: History and Everyday Sources
Table of Contents
- Abstract
- Introduction
- Chemical Identity and Properties
- Endocrine Disrupting Connection
- History of BPA: From Laboratory Curiosity to Global Contaminant
- Why BPA Became Indispensable: Industrial Applications
- Rise of BPA Analogues: BPS and BPF
- Common consumer sources
- Discussion and Conclusion: A Century of Unintended Consequences
- References
Abstract
Endocrine Disrupting Chemicals (EDCs) are a diverse class of exogenous substances that interfere with the endocrine system and cause adverse health effects in organisms or their progeny. Recognised as a significant global public health challenge, EDCs are ubiquitous in the modern environment due to their extensive use in industrial and consumer applications. This article synthesizes the formal definitions of EDCs, establishing a clear conceptual framework that distinguishes them from benign endocrine modulators. It further provides a comprehensive classification and analysis of major EDC sources, including plasticizers (e.g., Bisphenol A and phthalates), per- and polyfluoroalkyl substances (PFAS), industrial and agricultural pollutants (e.g., PCBs, dioxins, and pesticides), flame retardants, heavy metals, and environmental oestrogens. The pervasive presence of these chemicals in everyday products makes human exposure widespread and often unavoidable. The article underscores the critical need to address these pervasive contaminants to protect public health.
Introduction
Bisphenols are synthetic chemicals that have quietly woven themselves into the fabric of modern life. First synthesized in the late 19th century, compounds such as Bisphenol A (BPA) and its analogues Bisphenol S (BPS) and Bisphenol F (BPF) rapidly became indispensable to industry because of their durability, heat resistance, and ability to strengthen and clarify plastics [1; 2 ; 3 ; 4 ; 5 ; 6 ; 7]. Today, they are found in everyday items ranging from food containers and beverage bottles to receipts and electronics, making human exposure nearly unavoidable. These chemicals are classified as endocrine-disrupting chemicals (EDCs) because of their ability to interfere with hormonal signalling in humans and animals. To understand the scope of bisphenols' impact on modern life and human health, it is important to examine what they are chemically, how they came to be so ubiquitous, why attempts to replace them have largely failed, and ultimately why they remain a hidden but significant disruptor of human health and the environment.
Chemical Identity and Properties
Chemical Structure and Characteristics
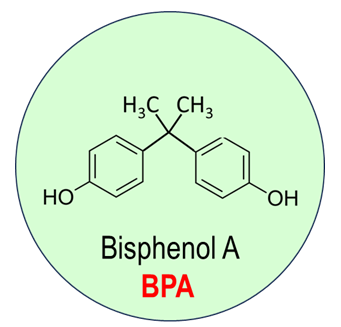
Bisphenol A, also known by its IUPAC name 4-[2-(4-hydroxyphenyl) propane-2yl] phenol, or 2,2-bis(4-hydroxyphenyl) propane, is an organic compound that belongs to the phenol group [4; 5; 6; 7; 8]. The "A" in its name is derived from acetone, one of its precursor materials [1].
Structurally, BPA is formed by two phenol groups linked by an isopropylidene group, with the chemical formula C₁₅H₁₆O₂ [4; 6; 7; 9; 10]. At room temperature, it is a white crystalline solid with a mild odour, low water solubility, but high fat solubility, nabling accumulation in biological tissues [11; 12]. Its lipophilic nature is a critical piece of the puzzle, as it explains how BPA can migrate from packaging into food and, once inside our bodies, potentially accumulate in fatty tissues. Figure 1 shows the chemical structure BPA.
Physical Properties with Biological Significance
BPA's physical properties help explain its persistence and biological impact. It has low solubility in water, approximately 200 mg/dm³ at 25 oC, but excellent solubility in fats, a lipophilic property indicated by its octanol-water partition coefficient (log Pow) of 3.32 to 3.8 [6; 9; 10; 11; 13]. Its fat solubility explains BPA's tendency to migrate from products into foods, especially fatty foods, and to accumulate in biological tissues [11; 12].
Endocrine Disrupting Connection
What makes BPA particularly concerning from a health perspective is its classification as a xenoestrogen. Its molecular structure, characterised by two hydroxyl groups in the para position, closely resembles both natural oestrogen (17β‑estradiol) and the synthetic oestrogen diethylstilbestrol (DES) [2; 5; 6; 14; 15]. This structural mimicry enables BPA to bind to oestrogen receptors (ERs), thereby interfering with normal hormonal signalling [2; 5; 11; 14]. Once bound, BPA can act as either an agonist or antagonist, initiating a cascade of endocrine disruption that underlies its wide range of adverse health effects [2; 5; 6; 14].
History of BPA: From Laboratory Curiosity to Global Contaminant
Initial Discovery and Synthesis (1891)
The story of BPA began in 1891, when Russian chemist Alexander P. Dianin first synthesized the compound by condensing acetone with two equivalents of phenol [1; 5; 6; 7; 10; 11; 13; 16; 17]. At that time, Dianin could not have foreseen that this laboratory creation would later become one of the most widely produced and environmentally pervasive synthetic chemicals in human history.
Estrogenic Revelation (1930s)
BPA remained largely overlooked until the 1930s, when its estrogenic properties were identified during the search for synthetic oestrogens by British chemist Charles Edward Dodds [1; 5; 6; 9; 13; 16]. This discovery revealed BPA’s ability to mimic natural hormones, a characteristic that would later prove both industrially valuable and biologically troubling. Notably, scientists recognised BPA’s hormonal activity decades before its widespread industrial adoption, yet this early knowledge did little to prevent its eventual ubiquity in consumer products.
Industrial Adoption and Mass Production (1950s–1960s)
The transformation of BPA from a laboratory curiosity into an industrial cornerstone occurred in the mid‑20th century. In the 1950s, researchers discovered that BPA could be polymerised into polycarbonate plastics, a material prized for its strength, transparency, and heat resistance [6]. This breakthrough opened the door to a wide range of consumer and industrial applications, from reusable beverage bottles and food containers to electronics and medical devices.
By the 1960s, BPA was being produced on a massive scale, marking the beginning of its global industrial dominance. Its versatility also extended to epoxy resins, which became essential for protective linings in food and beverage cans, water pipes, and dental composites. These dual applications, polycarbonates and epoxies, cemented BPA’s role as one of the most widely manufactured synthetic chemicals worldwide [4; 17; 18].
This period represents a turning point: BPA shifted from a chemical with recognised hormonal activity to a material foundation of modern consumer culture. Ironically, its industrial success unfolded despite earlier warnings about its estrogenic properties, setting the stage for decades of widespread exposure and subsequent health concerns.
Global Production Scale and Market Growth
Today, BPA is classified as a high‑production‑volume chemical, reflecting its central role in global manufacturing [10; 17; 19]. By 2013, worldwide annual production had reached approximately 6.7 million tons, and despite increasing regulatory scrutiny, output has consistently remained above 6.0 million tons in recent years [6; 9; 10; 16]. In 2022, global consumption was estimated at 7.7 million tons, with projections indicating continued growth as demand for plastics and resins expands [17].
These staggering figures highlight BPA’s deep entrenchment in modern industry. Its widespread use in polycarbonate plastics, epoxy resins, and thermal papers ensures that BPA is embedded in supply chains across packaging, consumer goods, electronics, and construction. This scale of production not only underscores its economic importance but also magnifies the scope of human and environmental exposure, raising urgent questions about sustainability, regulation, and safer alternatives.
Why BPA Became Indispensable: Industrial Applications
BPA achieved widespread adoption because of its unique combination of material properties, including elasticity, cross‑linking ability, heat resistance, and durability [2; 6; 9; 13; 20]. These characteristics made BPA not just another synthetic compound, but a cornerstone of modern manufacturing, enabling the production of materials that were stronger, clearer, and more resilient than many alternatives available at the time.
Its versatility positioned BPA as indispensable across three primary industrial domains, where it became the backbone of consumer plastics, epoxy resins, and thermal paper. Together, these applications explain why BPA remains one of the most produced chemicals worldwide and why its industrial entrenchment continues to challenge regulatory and health debates today.
Consumer Plastics
Nearly 70% of global BPA production is devoted to serving as a precursor for polycarbonate (PC) plastics [3]. These plastics quickly became embedded in everyday life, appearing in reusable beverage bottles, food storage containers, tableware, electronic components such as smartphone housings and cases, laptop shells, LED light covers, as well as in medical devices, and even children’s toys [1; 3; 5; 6; 8; 10; 13;19; 21].
The appeal of polycarbonate lay in its unique combination of clarity, strength, and heat resistance, which made it seem like a “miracle material” for consumer products. Unlike traditional plastics, PC offered glass‑like transparency without brittleness, enabling lightweight yet durable designs. Its resistance to high temperatures also made it suitable for applications requiring sterilisation, such as medical equipment, while its toughness ensured longevity in household goods and electronics.
By the late 20th century, PC had become a symbol of modern convenience and innovation, reinforcing BPA’s indispensability in global manufacturing. However, this widespread reliance also meant that BPA entered countless consumer pathways, setting the stage for the health and environmental concerns that continue to dominate discussions today.
Epoxy Resins: BPA’s Protective Shield
Beyond PCs, a significant share of BPA production is devoted to the manufacture of epoxy resins, which account for another major industrial application [6; 17]. Epoxy resins are prized for their adhesive strength, chemical resistance, and protective qualities, making them indispensable in coatings and linings.
One of their most widespread uses is in food and beverage packaging, where epoxy resins form the inner lining of cans and containers to prevent corrosion and contamination [ 8; 11; 22]. This innovation extended product shelf life and ensured consumer safety by keeping metals from leaching into food and drink. Epoxies also became critical in water pipes, construction materials, and dental composites, where their durability and resistance to moisture provided long‑lasting protection [1; 3; 4; 5; 6; 8; 10; 19; 20; 21].
By the mid‑20th century, epoxy resins had cemented BPA’s role in industries ranging from infrastructure to healthcare, reinforcing its reputation as a versatile chemical foundation. Yet, this protective shield came with unintended consequences: the same coatings that safeguarded products also became a direct pathway for BPA migration into food and water, intensifying human exposure.
Thermal Paper Applications
BPA also plays a critical role as a dye developer in thermal paper products, including cash register receipts, ATM slips, tickets, and labels [5; 6; 8; 10; 21; 23]. Unlike its polymerised form in plastics and resins, BPA in thermal paper exists in a free, unbound state on the paper’s surface. This makes it far more mobile and prone to direct transfer to human skin upon handling.
Because receipts are handled frequently and often discarded casually, this application represents a unique exposure pathway [11; 21]. Studies have shown that BPA can be absorbed dermally, particularly when contact is repeated or combined with conditions that enhance skin permeability, such as the use of hand sanitisers or lotions. Occupational exposure is of special concern for cashiers and retail workers, who may handle hundreds of receipts daily.
Although thermal paper accounts for a smaller fraction of BPA production compared to plastics and resins, its high potential for direct human contact has made it a focal point in regulatory debates Several regions have begun phasing out BPA‑based thermal paper in favor of substitutes, though questions remain about the safety of alternatives such as bisphenol S (BPS).
Rise of BPA Analogues: BPS and BPF
As scientific evidence of BPA's health effects accumulated and public concern grew, regulatory bodies began restricting its use in certain applications, particularly in products for infants and young children. This prompted manufacturers to develop and adopt structural analogues, primarily Bisphenol S (BPS) and Bisphenol F (BPF), resulting in a proliferation of products labeled "BPA-free" [1; 6; 7; 11; 15; 17; 24; 23; 25].
However, the safety of these substitutes has proven questionable, as they often exhibit similar or greater endocrine-disrupting activity compared to BPA [1; 11; 17; 25]. The replacement of one harmful chemical with structurally similar alternatives represents a pattern scientists have termed "regrettable substitution."
Bisphenol S
BPS (BPS; 4,4′‑sulphonyldiphenol) is a structural analogue of BPA that has been adopted across various industries as a presumed safer alternative [1; 17]. It is commonly found in PC plastics, the inner linings of food and beverage cans, and most notably, as a substitute for BPA in thermal papers [1; 10; 17]. Figure 2 shows the chemical structure of BFS.
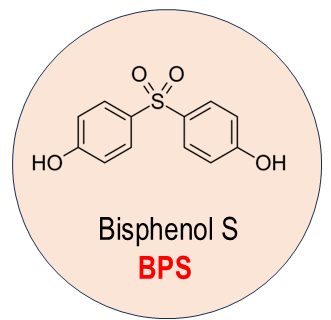
From a toxicity perspective, however, BPS is far from benign. Research shows that it possesses hormonal activity, including estrogenic, antiestrogenic, androgenic, and antiandrogenic effects [39]. While one in vitro assay suggested that BPS exhibits ten‑fold lower potency compared to BPA, other studies report comparable or even stronger endocrine‑disrupting effects, with evidence of obesogenic outcomes [1; 2; 6; 17].
This raises important concerns about consumer safety. The widespread marketing of “BPA‑free” products, particularly thermal receipts, may offer false reassurance, as substitution with BPS does not eliminate the risks associated with endocrine disruption. Instead, it highlights the broader challenge of regrettable substitution, where one hazardous chemical is replaced by another with similar or even greater toxicity.
Bisphenol F
BPF (BPF; 4,4′‑dihydroxydiphenylmethane) is another prominent structural analogue of bisphenol A. It is widely used in plastics and epoxy resins, and is particularly favoured in epoxy applications because it provides lower viscosity and greater solvent resistance compared to BPA [1; 17]. These properties have made BPF attractive to manufacturers seeking performance advantages or alternatives to BPA in industrial formulations. Figure 3 shows the chemical structure of BPF.
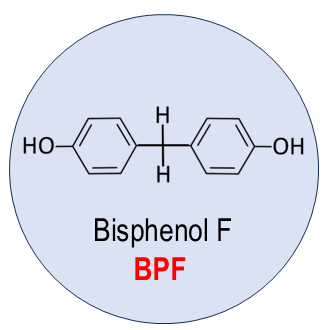
From a health perspective, however, BPF is recognised as an endocrine disruptor [1]. Its hormonal activity is comparable to BPA, with evidence of estrogenic, antiestrogenic, androgenic, and antiandrogenic effects [6; 7]. Alarmingly, some research suggests that BPF’s obesogenic potential may exceed that of BPA in preadipocyte models [1; 2] raising concerns that substitution with BPF could represent a “worse‑than‑the‑original” scenario.
This underscores the broader issue of regrettable substitution, where chemicals marketed as safer alternatives may carry equal or greater risks. Understanding BPF’s industrial role and biological activity is therefore critical to evaluating whether its adoption truly reduces harm or simply perpetuates the cycle of endocrine disruption.
Common consumer sources
Bisphenols such as BPA, BPS, and BPF are among the most pervasive EDCs. Human exposure occurs through ingestion (food and beverages), dermal contact, and inhalation, with consumer products serving as the primary pathways [6; 9; 26; 27; 28; 29; 30] {Figure 4). The following tables organise common sources of bisphenols into four major exposure categories.
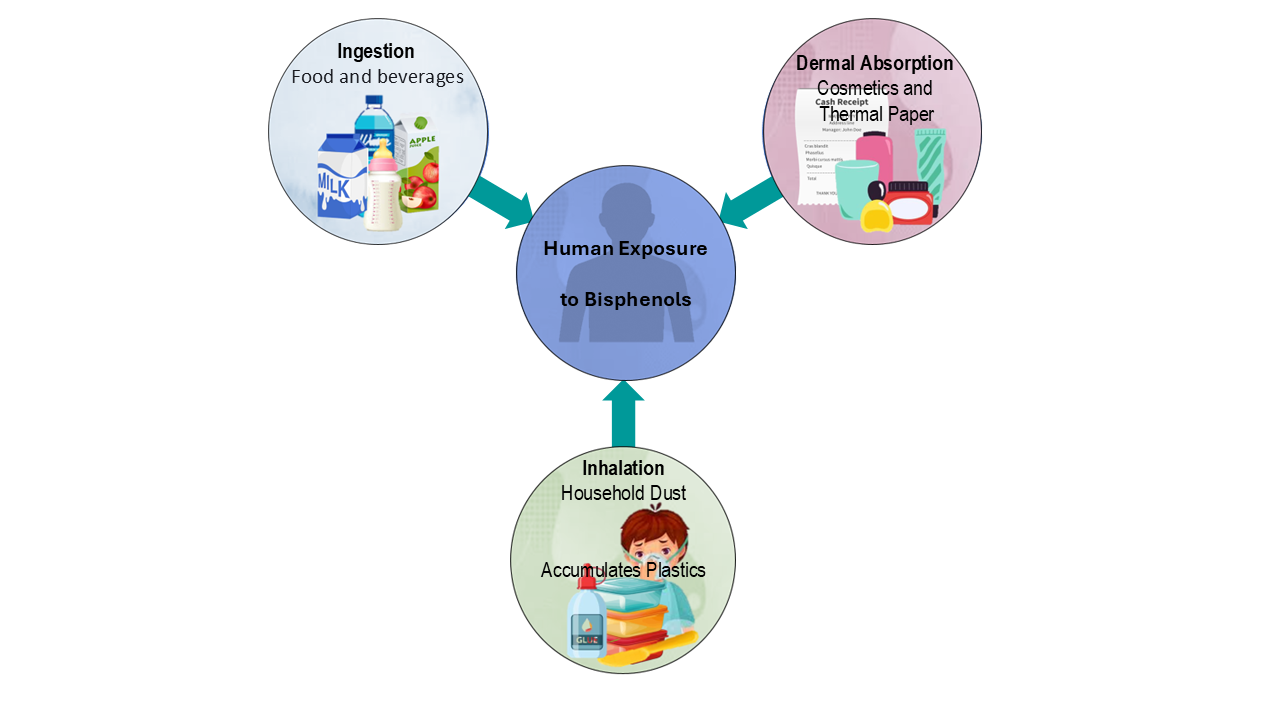
Food, Beverages, and Food Contact Materials (FCMs)
Food and beverage packaging represents the primary route of bisphenol exposure. BPA and its analogues leach from plastics and epoxy linings, especially when heated or reused [2; 5; 6; 7; 10; 29]. Table 1 shows bisphenol examples of food, beverages and food constact materials.
Table 1 Food, beverages, and food contact material bisphenol sources and examples
Bisphenol Source | Specific Consumer Examples | Citations |
Polycarbonate plastics | Reusable beverage bottles, water containers, baby bottles, tableware, food storage containers, microwave utensils, electronics (CDs/DVDs), eye lenses, safety equipment, children’s toys. | 4; 5; 7; 8; 10; 11; 12; 19; 22; 28; 31 |
Epoxy resins | Protective linings inside metal food and beverage cans, bottle tops. | 5; 7; 8; 22; 10; 11; 15; 19; 28; 32 |
Canned/processed foods | BPA/BPS/BPF leach from packaging, especially with heating or repeated use. | 5; 6; 8; 10; 11; 29; 30; 33 |
Dairy and seafood | BPA detected in canned dairy, cow’s milk, breast milk, freshwater fish, seafood. | 6; 9; 11; 25; 29; 34 |
Personal Care Products, Cosmetics, and Pharmaceuticals
Bisphenols are absorbed through dermal contact and incidental ingestion from personal care items. Thermal paper receipts are a particularly important source due to free BPA/BPS on the paper surface [26; 29; 31; 18]. Table 2 shows bisphenol examples of personal care products, cosmetics and pharmaceuticals.
Table 2 Personal care products, cosmetics and pharmaceutical bisphenol sources and examples
Bisphenol Source | Specific Consumer Examples | Citations |
Thermal paper | Cash register and ATM receipts, tickets, labels (major dermal exposure pathway). | 5; 8; 10; 11; 12; 29; 33; 35; 36 |
Cosmetics and perfumes | BPA/BPS occasionally detected in perfumes, lotions, and other cosmetic formulations. | 6; 10; 11 |
Household, Building Materials, and Indoor Environment
Indoor environments act as reservoirs for bisphenols, with dust accumulation and off‑gassing from plastics and papers contributing to chronic exposure [24; 29; 30; 33]. Table 3 shows household, building materials, and indoor environmental bisphenol examples.
Table 3 Household, bulding materials,and indoor environmental bisphenol sources and examples
Bisphenol Source | Specific Consumer Examples | Citations |
Household dust | BPA/BPS/BPF accumulate in indoor dust from plastics, electronics, and thermal paper. | 5; 6; 23; 24; 29 |
Building materials | Plastics, adhesives, and composites containing bisphenols contribute to indoor contamination. | 6; 29 |
Water and Environmental Contamination
Bisphenols enter water systems through industrial runoff, wastewater, and landfill leachate, contaminating drinking water and aquatic ecosystems. 9; 34]. Table 4 shows bisphenol sources and examples of water and environmental contamination.
Table 4 Water and environmental contamination bisphenol sources and examples
Bisphenol Source | Specific Consumer Examples | Citations |
Drinking water | Tap and bottled water contaminated via industrial runoff, landfill leachate, municipal wastewater, and supply pipes. | 5; 6; 9; 19; 26; 33; 35;37; 38 |
Soil and sludge | BPA/BPS/BPF enter soil through sewage sludge application and agricultural runoff. | 6; 34 |
Aquatic contamination | BPA detected in rivers, lakes, and sediments; bioaccumulates in fish and seafood. | 6; 9; 30 |
Discussion and Conclusion: A Century of Unintended Consequences
Taken together, BPA’s role in polycarbonate plastics, epoxy resins, and thermal papers explains why it became so deeply entrenched in modern industry. Its versatility allowed manufacturers to produce stronger, clearer, and longer‑lasting products, while its chemical properties enabled protective coatings and everyday consumer conveniences. Yet this very success ensured that BPA permeated countless aspects of daily life, creating widespread and often unavoidable exposure. The industrial indispensability of BPA thus stands in sharp contrast to the growing recognition of its health and environmental risks, a tension that continues to shape scientific inquiry and regulatory debate today.
The history of bisphenols represents a cautionary tale about industrial chemistry. Beginning with BPA’s synthesis in 1891 [1; 11] and its industrialization in the 1950s and 1960s [4; 6] we see a familiar pattern repeated across the chemical industry: the very properties that make a substance commercially valuable, durability, stability, and heat resistance, often also make it biologically persistent and potentially harmful. In the case of BPA, these traits ensured its dominance in consumer products but also magnified its capacity to disrupt endocrine systems and accumulate in the environment.
The widespread use of BPA and subsequent regulatory restrictions led to the adoption of alternatives such as BPS and BPF. Yet these substitutes, themselves endocrine‑disrupting chemicals with comparable risk profiles [1; 23], reveal the limitations of a substitution‑based approach. The “BPA‑free” label, while reassuring to consumers, often masks the reality of regrettable substitution, where one hazardous compound is simply replaced by another with similar or even greater toxicity. This pattern underscores that addressing endocrine disruption requires more than swapping one bisphenol for another. It demands a fundamental rethinking of how chemicals are designed, tested, and regulated before they become embedded in millions of consumer products, and ultimately, in our bodies.
References
- Biemann, R., Blüher, M., & Isermann, B. (2021). Exposure to endocrine-disrupting compounds such as phthalates and bisphenol A is associated with an increased risk for obesity. Best Practice & Research Clinical Endocrinology & Metabolism, 35, 101546. https://doi.org/10.1016/j.beem.2021.101546
- Dalamaga, M., Kounatidis, D., Tsilingiris, D., Vallianou, N. G., Karampela, I., Psallida, S., & Papavassiliou, A. G. (2024). The role of endocrine disruptors bisphenols and phthalates in obesity: Current evidence, perspectives and controversies. International Journal of Molecular Sciences, 25, 675. https://doi.org/10.3390/ijms25010675
- Fu, X., Yana, R., Zhou, G., Chen, X., Liu, Y., Chi, J., Li, X., Fang, H., Li, H., & Li, W. (2022). New progress in photocatalytic degradation of bisphenol A as representative endocrine disrupting chemicals. Current Opinion in Green and Sustainable Chemistry, 35, 100629. https://doi.org/10.1016/j.cogsc.2022.100629
- Gałązka, A., & Jankiewicz, U. (2022). Endocrine disrupting compounds (nonylphenol and bisphenol A)–Sources, harmfulness and laccase-assisted degradation in the aquatic environment. Microorganisms, 10, 2236. https://doi.org/10.3390/microorganisms10112236
- Kawa, I. A., Masood, A., Fatima, Q., Mir, S. A., Jeelani, H., Manzoor, S., & Rashid, F. (2021). Endocrine disrupting chemical bisphenol A and its potential effects on female health. Diabetes & Metabolic Syndrome: Clinical Research & Reviews, 15, 803–811. https://doi.org/10.1016/j.dsx.2021.03.031
- Presunto, M., Mariana, M., Lorigo, M., & Cairrao, E. (2023). The effects of bisphenol A on human male infertility: A review of current epidemiological studies. International Journal of Molecular Sciences, 24, 12417. https://doi.org/10.3390/ijms241512417
- Salami, E. A., & Rotimi, O. A. (2024). The impact of bisphenol-A on human reproductive health. Toxicology Reports, 13, 101773. https://doi.org/10.1016/j.toxrep.2024.101773
- Czarnywojtek, A., Jaz, K., Ochmańska, J., Zgorzalewicz-Stachowiak, M., Czarnocka, B., Sawicka-Gutaj, N., Ziółkowska, P., Krela-Kaźmierczak, I., Gut, P., Florek, E., & Ruchała, M. (2021). The effect of endocrine disruptors on the reproductive system – Current knowledge. European Review for Medical and Pharmacological Sciences, 25, 4930–4940.
- Costa, H. E., & Cairrao, E. (2024). Effect of bisphenol A on the neurological system: A review update. Archives of Toxicology, 98, 1–73. https://doi.org/10.1007/s00204-023-03614-0
- Tsai, W.-T. (2023). Survey on the environmental risks of bisphenol A and its relevant regulations in Taiwan: An environmental endocrine-disrupting chemical of increasing concern. Toxics, 11, 722. https://doi.org/10.3390/toxics11090722
- Buoso, E., Masi, M., Limosani, R. V., Oliviero, C., Saeed, S., Iulini, M., Passoni, F. C., Racchi, M., & Corsini, E. (2025). Endocrine disrupting toxicity of bisphenol A and its analogs: Implications in the neuro-immune milieu. Journal of Xenobiotics, 15, 13. https://doi.org/10.3390/jox15010013
- Manzan-Martins, C., & Paulesu, L. (2021). Impact of bisphenol A (BPA) on cells and tissues at the human materno-fetal interface. Tissue and Cell, 73, 101662. https://doi.org/10.1016/j.tice.2021.101662
- Fonseca, M. I., Lorigo, M., & Cairrao, E. (2022). Endocrine-disrupting effects of bisphenol A on the cardiovascular system: A review. Journal of Xenobiotics, 12, 181–213. https://doi.org/10.3390/jox12030015
- Palacios-Arreola, M. I., Moreno-Mendoza, N. A., Nava-Castro, K. E., Segovia-Mendoza, M., Perez-Torres, A., Garay-Canales, C. A., & Morales-Montor, J. (2022). The endocrine disruptor compound bisphenol-A (BPA) regulates the intra-tumoral immune microenvironment and increases lung metastasis in an experimental model of breast cancer. International Journal of Molecular Sciences, 23, 2523. https://doi.org/10.3390/ijms23052523
- Rifa, R. A., & Lavado, R. (2024). Cytotoxic impacts of seven alternative bisphenols on human in vitro cellular models. Chemosphere, 366, 143408. https://doi.org/10.1016/j.chemosphere.2024.143408
- Buha, A., Manic, L., Maric, D., Tinkov, A., Skolny, A., Antonijevic, B., & Hayes, A. W. (2022). The effects of endocrine-disrupting chemicals (EDCs) on the epigenome—A short overview. Toxicology Research and Application, 6, 1–9. https://doi.org/10.1177/23978473221115817
- Stanojević, M., & Sollner Dolenc, M. (2025). Mechanisms of bisphenol A and its analogs as endocrine disruptors via nuclear receptors and related signaling pathways. Archives of Toxicology, 99, 2397–2417. https://doi.org/10.1007/s00204-025-04025-z
- Zhang, X., Flaws, J. A., Spinella, M. J., & Irudayaraj, J. (2023). The relationship between typical environmental endocrine disruptors and kidney disease. Toxics, 11, 32. https://doi.org/10.3390/toxics11010032
- Gonsioroski, A., Mourikes, V. E., & Flaws, J. A. (2020). Endocrine disruptors in water and their effects on the reproductive system. International Journal of Molecular Sciences, 21(6), 1929. https://doi.org/10.3390/ijms21061929
- Palsania, P., Singhal, K., Dar, M. A., & Kaushik, G. (2024). Food grade plastics and Bisphenol A: Associated risks, toxicity, and bioremediation approaches. Journal of Hazardous Materials, 466, 133474. https://doi.org/10.1016/j.jhazmat.2024.133474
- Adegoke, E. O., Rahman, M. S., Amjad, S., Pang, W.-K., Ryu, D.-Y., Park, Y.-J., & Pang, M.-G. (2022). Bisphenol A damages testicular junctional proteins transgenerationally in mice. Environmental Pollution, 302, 119067. https://doi.org/10.1016/j.envpol.2022.119067
- Andonotopo, W., Bachnas, M. A., Dewantiningrum, J., Pramono, M. B. A., Akbar, M. I. A., Darmawan, E., Sanjaya, N. H., Aldiansyah, D., Sulistyowati, S., Stanojevic, M., & Kurjak, A. (2025). Endocrine disrupting chemicals: Translating mechanisms into perinatal risk assessment. Journal of Perinatal Medicine. Advance online publication. https://doi.org/10.1515/jpm-2025-0259
- Seewoo, B. J., Wong, E. V. S., Mulders, Y. R., Gozt, A., Elagali, A., Symeonides, C., & Dunlop, S. A. (2024). A systematic evidence map protocol for mapping global exposure to bisphenols and their alternatives and social and environmental justice implications. Environment International, 194, 109091. https://doi.org/10.1016/j.envint.2024.109091
- Land, K. L., Miller, F. G., Fugate, A. C., & Hannon, P. R. (2022). The effects of endocrine-disrupting chemicals on ovarian- and ovulation-related fertility outcomes. Molecular Reproduction and Development, 89(6), 608–631.
- Pathak, R. K., Jung, D.-W., Shin, S.-H., Ryu, B.-Y., Lee, H.-S., & Kim, J.-M. (2024). Deciphering the mechanisms and interactions of the endocrine disruptor bisphenol A and its analogs with the androgen receptor. Journal of Hazardous Materials, 469, 133935. https://doi.org/10.1016/j.jhazmat.2024.133935
- Bertram, M. G., Gore, A. C., Tyler, C. R., & Brodin, T. (2002). Endocrine-disrupting chemicals. World Health Organization. https://apps.who.int/iris/handle/10665/67357
- Di Pietro, G., Forcucci, F., & Chiarelli, F. (2023). Endocrine disruptor chemicals and children’s health. International Journal of Molecular Sciences, 24(3), 2671. https://doi.org/10.3390/ijms24032671
- Ghosh, A., Tripathy, A., & Ghosh, D. (2022). Impact of endocrine disrupting chemicals (EDCs) on reproductive health of humans. Proceedings of the Zoological Society, 75(1), 16–30. https://doi.org/10.1007/s12595-021-00412-3
- Goralczyk, K. (2021). A review of the impact of selected anthropogenic chemicals from the group of endocrine disruptors on human health. Toxics, 9, 146. https://doi.org/10.3390/toxics9070146
- Ma, C.-X., Ma, X.-N., Li, H.-L., Mauricio, D., & Fu, S.-B. (2025). Endocrine-disrupting chemicals exposure: Cardiometabolic health risk in humans. Cardiovascular Diabetology, 24, 381. https://doi.org/10.1186/s12933-025-02938-8
- Park, S. Y., Kong, S. H., Kim, K. J., Ahn, S. H., Hong, N., Ha, J., Lee, S., Choi, H. S., Baek, K.-H., Kim, J.-E., & Kim, S. W. (2024). Effects of endocrine-disrupting chemicals on bone health. Endocrinology and Metabolism, 39, 539–551. https://doi.org/10.3803/EnM.2024.1963
- Peters, A. E., Ford, E. A., Roman, S. D., Bromfield, E. G., Nixon, B., Pringle, K. G., & Sutherland, J. M. (2024). Impact of bisphenol A and its alternatives on oocyte health: A scoping review. Human Reproduction Update, 30(6), 653–691. https://doi.org/10.1093/humupd/dmae025
- Trasande, L., & Sargis, R. M. (2024). Endocrine-disrupting chemicals: Mainstream recognition of health effects and implications for the practicing internist. Journal of Internal Medicine, 295(2), 259–274. https://doi.org/10.1111/joim.13748
- Martín-Pozo, L., Mejías, C., Santos, J. L., Martín, J., Aparicio, I., & Alonso, E. (2024). Influence of microplastic contamination on the dissipation of endocrine disrupting chemicals in soil environment. Environmental Pollution, 349, 123919. https://doi.org/10.1016/j.envpol.2024.123919
- Hall, J. M., & Greco, C. W. (2020). Perturbation of nuclear hormone receptors by endocrine disrupting chemicals: Mechanisms and pathological consequences of exposure. Cells, 9(1), 13. https://doi.org/10.3390/cells9010013
- Virant-Klun, I., Imamovic-Kumalic, S., & Pinter, B. (2022). From oxidative stress to male infertility: Review of the associations of endocrine-disrupting chemicals (bisphenols, phthalates, and parabens) with human semen quality. Antioxidants, 11, 1617. https://doi.org/10.3390/antiox11081617
- Farounbi, A. I., & Ngqwala, N. P. (2020). Occurrence of selected endocrine disrupting compounds in the Eastern Cape Province of South Africa. Environmental Science and Pollution Research, 27, 17268–17279. https://doi.org/10.1007/s11356-020-08082-y
- Mokra, K. (2021). Endocrine disruptor potential of short- and long-chain perfluoroalkyl substances (PFASs)—A synthesis of current knowledge with proposal of molecular mechanism. International Journal of Molecular Sciences, 22(4), 2148. https://doi.org/10.3390/ijms22042148
- Kek, T., Geršak, K., & Virant‑Klun, I. (2024). Exposure to endocrine disrupting chemicals (bisphenols, parabens, and triclosan) and their associations with preterm birth in humans. Reproductive Toxicology, 125, 108580. https://doi.org/10.1016/j.reprotox.2024.108580
