Endocrine Disrupting Chemicals: A Global Threat to Health and Development

Table of Contents
Abstract
Endocrine Disrupting Chemicals (EDCs) represent a pervasive and escalating threat to global public health. These exogenous substances or mixtures alter the function of the endocrine system and consequently cause adverse health effects in intact organisms, their progeny, or (sub)populations. They are implicated in a wide array of biological disruptions and challenge traditional toxicological paradigms. This introductory article synthesizes the current scientific literature on the mechanisms, exposure pathways, vulnerable populations, and resultant outcomes associated with EDCs, establishing the foundation for deeper thematic exploration of this critical environmental health challenge.
Introduction
The recognition of Endocrine Disrupting Chemicals (EDCs) as a critical global health concern, is growing. There is mounting evidence linking them to adverse outcomes across reproductive, metabolic, thyroid, and neurodevelopmental domains (1; 2). These chemicals interfere with hormonal signalling and regulation, often at exceptionally low doses and through complex mixtures, fundamentally challenging traditional toxicological paradigms that assume "the dose makes the poison" (3; 4; 5; 6; 7). The ubiquity of EDCs in modern society, combined with their ability to exert effects across generations through epigenetic mechanisms, positions them as a unique environmental health challenge requiring urgent scientific and regulatory attention.
Exposure Pathways and Population Vulnerability
Sources and Routes of Exposure
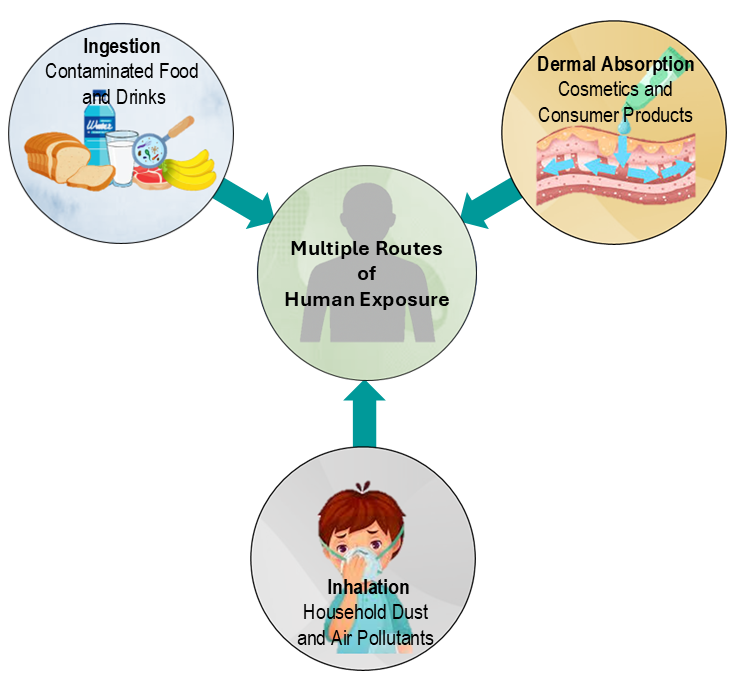
EDCs are ubiquitous in modern environments, contaminating our daily lives through their presence in plastics, food packaging, water supplies, soil, cosmetics, and industrial waste (3; 8; 9). Human exposure occurs through multiple routes: ingestion (contaminated food and water), inhalation (household dust and air pollutants), and dermal absorption (cosmetics and consumer products) (5; 10) (Figure 1).
Vulnerable Populations and Critical Windows
Critical Windows of Exposure: Prenatal and early childhood periods represent windows of heightened vulnerability to EDC exposure. During these critical developmental phases, even low-level exposures can produce profound and irreversible effects that may not manifest until adolescence or adulthood, a phenomenon known as "latency" (1; 3; 6; 10; 11; 12; 13).
Health Disparities: Marginalised and low-income populations frequently face disproportionate exposure due to factors such as substandard housing, including residential proximity to industrial sites, occupational hazards, and limited access to uncontaminated food and water. This environmental disparity exacerbates existing health inequalities and perpetuates cycles of disadvantage (2; 14).
Mechanisms of Endocrine Disruption
EDCs exert their effects through several interconnected molecular pathways that disrupt normal endocrine function.
Hormone Mimicry and Receptor Interference
Many endocrine-disrupting chemicals (EDCs) are structural analogues of endogenous hormones, particularly oestrogens and androgens. For example, bisphenol A (BPA) closely mimics the molecular structure of oestradiol, enabling it to bind to oestrogen receptors and disrupt normal hormonal signalling. These chemicals can act either as agonists (activating the receptor) or antagonists (blocking natural hormone binding). This molecular mimicry can trigger inappropriate cellular responses or prevent normal hormonal signalling (4; 5; 6; 9; 10; 11; 13; 15; 16).
Disruption of Hormone Homeostasis
Beyond receptor interactions, EDCs interfere with the entire lifecycle of hormone regulation. They can inhibit or enhance hormone synthesis in endocrine glands, alter hormone transport through the bloodstream by binding to carrier proteins, modify hormone metabolism in the liver and other tissues, and affect hormone clearance and excretion. These disruptions compromise the delicate balance required for normal endocrine function (1; 6; 9; 10; 14; 17; 18).
Epigenetic Modifications
A particularly concerning aspect of EDC toxicity is their ability to induce epigenetic changes, alterations in gene expression without changes to the underlying DNA sequence. EDCs can modify DNA methylation patterns, alter histone structure and function, and disrupt non-coding RNA expression. These epigenetic modifications can persist throughout an individual's lifetime and, critically, may be transmitted across generations, creating transgenerational health effects (3; 5; 6; 13; 14; 19; 20).
Non-Monotonic Dose Responses and Mixture Effects
EDCs frequently exhibit non-monotonic dose-response relationships, wherein low doses can produce effects equal to or greater than those observed at higher doses. This phenomenon contradicts classical toxicological assumptions and has profound implications for risk assessment and regulatory thresholds. Additionally, humans are rarely exposed to single EDCs in isolation. The "cocktail effect" of combined exposures may produce additive or synergistic toxicity, even when individual chemicals are present at concentrations below their supposed "safe" levels (6; 9; 11; 20).
Health Impacts
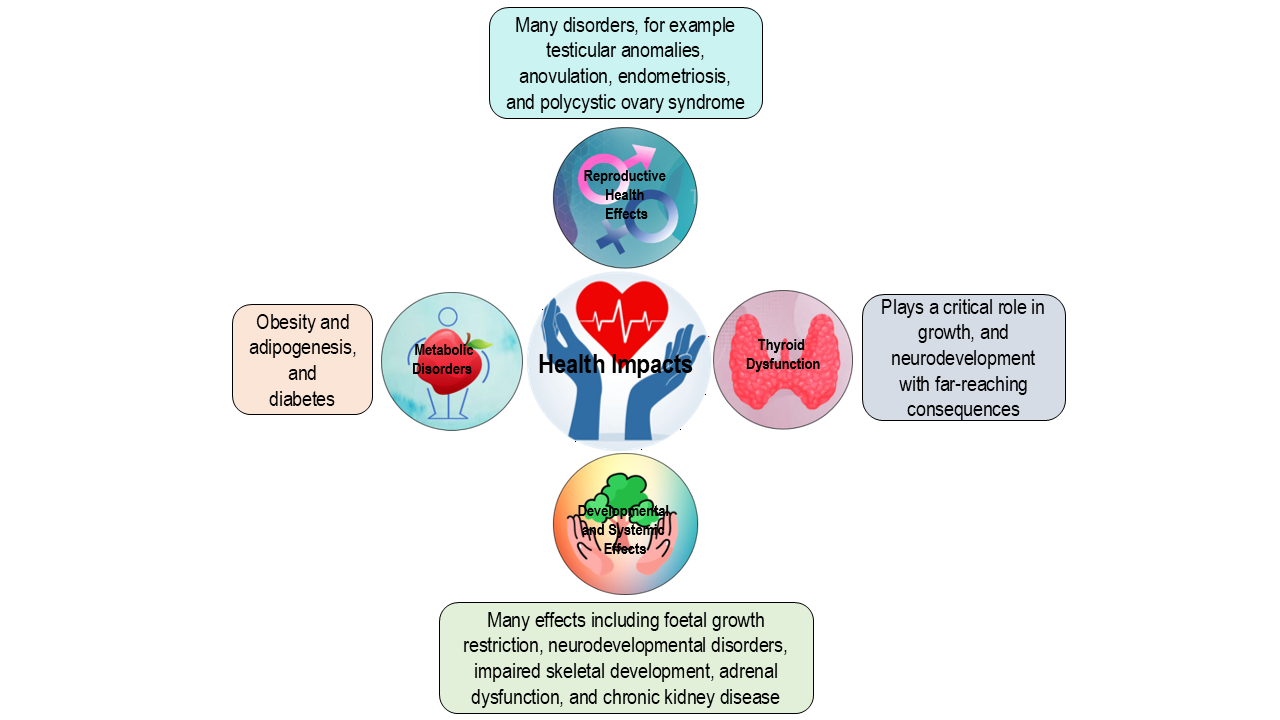
EDCs interfere with the body’s hormone system, and even small exposures can have wide‑ranging health impacts. They are linked to many health problems including reproductive, thyroid, and metabolic disorders, as well as wide ranging growth and developmental effects (Figure 2).
Reproductive Health
EDCs disrupt the Hypothalamic-Pituitary-Gonadal (HPG) axis, the central regulatory system governing reproductive function, leading to impaired fertility and abnormal sexual development (15; 17; 21; 22).
Male Reproductive Effects: EDC exposure is linked to reduced sperm quality (decreased count, motility, and morphology), testicular anomalies including cryptorchidism and hypospadias, and decreased testosterone production. Notably, PFOA (perfluorooctanoic acid) exposure is associated with increased testicular cancer risk (3; 5; 6; 10).
Female Reproductive Effects: Bisphenol A (BPA) and per- and polyfluoroalkyl substances (PFAS) are associated with anovulation, endometriosis, and polycystic ovary syndrome (PCOS). Historical exposure to diethylstilbestrol (DES) during pregnancy has been linked to reproductive tract cancers in female offspring, demonstrating the severe consequences of developmental EDC exposure (3; 10; 11; 15; 17; 22).
Metabolic Disorders
EDCs contribute significantly to the global epidemic of metabolic disease, including obesity, insulin resistance, and Type 2 Diabetes Mellitus (T2DM) (6; 7; 10; 11; 15; 19).
Obesity and Adipogenesis: Early-life exposure to bisphenols and phthalates promotes adipocyte differentiation and increases adiposity, predisposing individuals to lifelong metabolic dysfunction. These chemicals act as "obesogens," reprogramming metabolic pathways during critical developmental windows (7; 11; 12; 18).
Diabetes: BPA and PFAS exposure are linked to T2DM and gestational diabetes through multiple mechanisms, including pancreatic beta-cell dysfunction and the induction of insulin resistance in peripheral tissues (19; 23; 24).
Thyroid Dysfunction
The thyroid gland is particularly sensitive to EDC disruption. Chemicals such as polychlorinated biphenyls (PCBs), PFAS, and brominated flame retardants interfere with thyroid hormone synthesis, disrupt thyroid hormone transport proteins, and alter peripheral metabolism of thyroid hormones. Given the critical role of thyroid hormones in metabolism, growth, and neurodevelopment, these disruptions have far-reaching consequences (3; 10; 25; 26; 27).
Developmental and Systemic Effects
The health impacts of EDCs extend beyond the classical endocrine targets to affect multiple organ systems and developmental outcomes. Documented effects include preterm birth, foetal growth restriction, neurodevelopmental disorders (including autism spectrum disorder), impaired bone mineralization and skeletal development, adrenal dysfunction affecting stress responses, and chronic kidney disease (2; 13; 14; 18; 20; 22; 28; 29; 30).
These systemic effects underscore that EDCs should be understood not merely as endocrine disruptors, but as agents capable of perturbing multiple interconnected physiological systems.
Policy and Research Imperatives
Regulatory Challenges
Despite robust epidemiological and experimental evidence documenting EDC harms, regulatory action remains inadequate and fragmented across jurisdictions. Chemical regulation has historically relied on single-chemical risk assessment and assumed monotonic dose-response relationships; frameworks poorly suited to EDC toxicology.
The Precautionary Principle
The precautionary principle, which holds that action to prevent harm should not await scientific certainty, should guide EDC policy, particularly to protect vulnerable populations including pregnant women, infants, and children. This approach justifies regulatory action even in the face of scientific uncertainty, prioritizing prevention over remediation (2; 11; 19).
Individual Risk Reduction
While systemic policy change is essential, individuals can take steps to reduce personal EDC exposure. Recommended strategies include avoiding plastic food containers and water bottles (particularly those containing BPA), choosing fresh or frozen foods over canned goods, scrutinizing personal care products for phthalates and parabens, using HEPA filters to reduce indoor air contamination, and selecting EDC-free household products when possible (3; 6; 19; 23).
Future Research Priorities
Research priorities must evolve to reflect real-world exposure scenarios. Critical needs include longitudinal cohort studies tracking exposures from prenatal life through adulthood, mixture toxicity studies evaluating cocktail effects, mechanistic research elucidating epigenetic pathways and transgenerational effects, and improved biomonitoring methods for emerging EDCs (3; 6; 19; 23).
Conclusion
Endocrine Disrupting Chemicals represent a fundamental challenge to environmental health in the 21st century. Their ubiquity, mechanistic complexity, transgenerational effects, and capacity to disrupt multiple physiological systems demand coordinated action from researchers, policymakers, healthcare providers, and the public. Addressing the EDC crisis requires not only advancing scientific understanding but also reimagining chemical regulation, environmental justice, and the precautionary frameworks that should guide public health protection.
References
Cortes-Ramirez, S. A., Ho, S.-M., & Leung, Y.-K. (2024). Endocrine-disrupting chemicals: A looming threat to current and future generations. International Journal of Molecular Sciences, 25(15), 8222. https://doi.org/10.3390/ijms25158222
- Trasande, L., & Sargis, R. M. (2024). Endocrine-disrupting chemicals: Mainstream recognition of health effects and implications for the practicing internist. Journal of Internal Medicine, 295(2), 259–274. https://doi.org/10.1111/joim.13748
- Alsen, M., Sinclair, C., Cooke, P., Ziadkhanpour, K., Genden, E., & van Gerwen, M. (2021). Endocrine disrupting chemicals and thyroid cancer: An overview. Toxics, 9(1), 14. https://doi.org/10.3390/toxics9010014
- Bonato, M., Corrà, F., Bellio, M., Guidolin, L., Tallandini, L., Irato, P., & Santovito, G. (2020). PFAS environmental pollution and antioxidant responses: An overview of the impact on human field. International Journal of Environmental Research and Public Health, 17(21), 8020. https://doi.org/10.3390/ijerph17218020
- Cargnelutti, F., Di Nisio, A., Pallotti, F., Sabovic, I., Spaziani, M., Tarsitano, M. G., Paoli, D., & Foresta, C. (2021). Effects of endocrine disruptors on fetal testis development, male puberty, and transition age. Endocrine, 72, 358–374. https://doi.org/10.1007/s12020-020-02436-9
- Di Pietro, G., Forcucci, F., & Chiarelli, F. (2023). Endocrine disruptor chemicals and children’s health. International Journal of Molecular Sciences, 24(3), 2671. https://doi.org/10.3390/ijms24032671
- Haverinen, E., Fernandez, M. F., Mustieles, V., & Tolonen, H. (2021). Metabolic syndrome and endocrine disrupting chemicals: An overview of exposure and health effects. International Journal of Environmental Research and Public Health, 18(24), 13047. https://doi.org/10.3390/ijerph182413047
- Gonsioroski, A., Mourikes, V. E., & Flaws, J. A. (2020). Endocrine disruptors in water and their effects on the reproductive system. International Journal of Molecular Sciences, 21(6), 1929. https://doi.org/10.3390/ijms21061929
- Leung, Y.-K. (2023). A silent threat: Exploring the impact of endocrine disruption on human health. International Journal of Molecular Sciences, 24(12), 9790. https://doi.org/10.3390/ijms24129790
- Guarnotta, V., Amodei, R., Frasca, F., Aversa, A., & Giordano, C. (2022). Impact of chemical endocrine disruptors and hormone modulators on the endocrine system. International Journal of Molecular Sciences, 23(10), 5710. https://doi.org/10.3390/ijms23105710
- Hall, J. M., & Greco, C. W. (2020). Perturbation of nuclear hormone receptors by endocrine disrupting chemicals: Mechanisms and pathological consequences of exposure. Cells, 9(1), 13. https://doi.org/10.3390/cells9010013
- Pezeshki, H., Rajabi, S., Hashemi, M., Moradalizadeh, S., & Nasab, H. (2025). Per- and poly-fluoroalkyl substances as forever chemicals in drinking water: Unraveling the nexus with obesity and endocrine disruption – A mini review. Heliyon, 11, e42782. https://doi.org/10.1016/j.heliyon.2025.e42782
- Shulhai, A.-M., Palanza, P., & Street, M. E. (2024). Current evidence on the effects of endocrine-disrupting chemicals (EDCs) on bone growth and health. Exposure and Health, 16, 1001–1025. https://doi.org/10.1007/s12403-023-00607-3
- Andonotopo, W., Bachnas, M. A., Dewantiningrum, J., Pramono, M. B. A., Akbar, M. I. A., Darmawan, E., Sanjaya, N. H., Aldiansyah, D., Sulistyowati, S., Stanojevic, M., & Kurjak, A. (2025). Endocrine disrupting chemicals: Translating mechanisms into perinatal risk assessment. Journal of Perinatal Medicine. Advance online publication. https://doi.org/10.1515/jpm-2025-0259
- Land, K. L., Miller, F. G., Fugate, A. C., & Hannon, P. R. (2022). The effects of endocrine-disrupting chemicals on ovarian- and ovulation-related fertility outcomes. Molecular Reproduction and Development, 89(6), 608–631. https://doi.org/10.1002/mrd.23652
- Mokra, K. (2021). Endocrine disruptor potential of short- and long-chain perfluoroalkyl substances (PFASs)—A synthesis of current knowledge with proposal of molecular mechanism. International Journal of Molecular Sciences, 22(4), 2148. https://doi.org/10.3390/ijms22042148
- Voros, C., Athanasiou, D., Papapanagiotou, I., Mavrogianni, D., Varthaliti, A., Bananis, K., Athanasiou, A., Papadimas, G., Gkirgkinoudis, A., et al. (2025). Molecular shadows of per- and polyfluoroalkyl substances (PFASs): Unveiling the impact of perfluoroalkyl substances on ovarian function, polycystic ovarian syndrome (PCOS), and in vitro fertilization (IVF) outcomes. International Journal of Molecular Sciences, 26(6604). https://doi.org/10.3390/ijms26146604
- Zhang, X., Flaws, J. A., Spinella, M. J., & Irudayaraj, J. (2023). The relationship between typical environmental endocrine disruptors and kidney disease. Toxics, 11(32). https://doi.org/10.3390/toxics11010032
- Ma, C.-X., Ma, X.-N., Li, H.-L., Mauricio, D., & Fu, S.-B. (2025). Endocrine-disrupting chemicals exposure: Cardiometabolic health risk in humans. Cardiovascular Diabetology, 24, 381. https://doi.org/10.1186/s12933-025-02938-8
- Stüfchen, I., Schweizer, J. R. O. L., Völter, F., Nowak, E., Braun, L., Kocabiyik, J., Mederos y Schnitzler, M., Williams, T. A., Kunz, S., Bidlingmaier, M., & Reincke, M. (2025). The impact of endocrine disrupting chemicals on adrenal corticosteroids – A systematic review of epidemiological studies. Environmental Research, 276, 121438. https://doi.org/10.1016/j.envres.2025.121438
- Nguyen, T., Nsiah, G. A., Crowder, E., Garland, S., Williams, C. F., & Conroy-Ben, O. (2024). Predicted endocrine disrupting activity of unregulated drinking water contaminants. ACS ES&T Water, 4(4), 1000–1013. https://doi.org/10.1021/acsestwater.3c00156
- Rickard, B. P., Rizvi, I., & Fenton, S. E. (2022). Per- and poly-fluoroalkyl substances (PFAS) and female reproductive outcomes: PFAS elimination, endocrine-mediated effects, and disease. Toxicology, 465, 153031. https://doi.org/10.1016/j.tox.2021.153031
- Onyebuchi, C., Muogbo, I., Chukwurah, C. O., Abdulkadir, M., Gordon, O. P., Ogundare, S. A., & Agaba, I. (2025). [Article title not provided]. medtigo Journal of Pharmacology, 2(3). https://doi.org/10.63096/medtigo30612310
- Yan, D., Jiao, Y., Yan, H., Liu, T., Yan, H., & Yuan, J. (2022). Endocrine-disrupting chemicals and the risk of gestational diabetes mellitus: A systematic review and meta-analysis. Environmental Health, 21, 53. https://doi.org/10.1186/s12940-022-00858-8
- Coperchini, F., Croce, L., Ricci, G., Magri, F., Rotondi, M., Imbriani, M., & Chiovato, L. (2021). Thyroid disrupting effects of old and new generation PFAS. Frontiers in Endocrinology, 11, 612320. https://doi.org/10.3389/fendo.2020.612320
- Panieri, E., Buha Đorđevic, A., & Saso, L. (2021). Endocrine disruption by PFAS: A major concern associated with legacy and replacement substances. Arhiv za farmaciju, 71, 429–454. https://doi.org/10.5937/arhfarm71-34197
- Sun, M., Cao, X., Wu, Y., Shen, L., & Wei, G. (2022). Prenatal exposure to endocrine-disrupting chemicals and thyroid function in neonates: A systematic review and meta-analysis. Ecotoxicology and Environmental Safety, 231, 113215. https://doi.org/10.1016/j.ecoenv.2022.113215
- Derakhshan, A., Shu, H., Broeren, M. A. C., Kortenkamp, A., Lindh, C. H., Demeneix, B., Peeters, R. P., Bornehag, C.-G., & Korevaar, T. I. M. (2023). Association of endocrine disrupting chemicals exposure with human chorionic gonadotropin concentrations in pregnancy. Environment International, 178, 108091. https://doi.org/10.1016/j.envint.2023.108091
- Iwobi, N., & Sparks, N. R. (2023). Endocrine disruptor-induced bone damage due to hormone dysregulation: A review. International Journal of Molecular Sciences, 24(8263). https://doi.org/10.3390/ijms24098263
- Vuong, A. M., Braun, J. M., Sjödin, A., Calafat, A. M., Yolton, K., Lanphear, B. P., & Chen, A. (2021). Exposure to endocrine disrupting chemicals (EDCs) and cardiometabolic indices during pregnancy: The HOME Study. Environment International, 156, 106747. https://doi.org/10.1016/j.envint.2021.106747
