Endocrine Disrupting Chemicals: Definition and Sources

Table of Contents
Abstract
Endocrine Disrupting Chemicals (EDCs) are a diverse class of exogenous substances that interfere with the endocrine system and cause adverse health effects in organisms or their progeny. Recognised as a significant global public health challenge, EDCs are ubiquitous in the modern environment due to their extensive use in industrial and consumer applications. This article synthesizes the formal definitions of EDCs, establishing a clear conceptual framework that distinguishes them from benign endocrine modulators. It further provides a comprehensive classification and analysis of major EDC sources, including plasticizers (e.g., Bisphenol A and phthalates), per- and polyfluoroalkyl substances (PFAS), industrial and agricultural pollutants (e.g., PCBs, dioxins, and pesticides), flame retardants, heavy metals, and environmental oestrogens. The pervasive presence of these chemicals in everyday products makes human exposure widespread and often unavoidable. The article underscores the critical need to address these pervasive contaminants to protect public health.
Introduction
The ubiquitous presence of synthetic and natural compounds capable of interfering with the body's endocrine signalling system has emerged as a significant global public health concern. These substances, collectively termed Endocrine Disrupting Chemicals (EDCs), represent a diverse class of environmental contaminants with the capacity to perturb hormonal homeostasis at multiple physiological levels [1; 2; 3; 4; 5]. The recognition of EDCs as a critical environmental health challenge has intensified over the past two decades, driven by accumulating evidence of their biological activity even at low environmental concentrations and their persistence in biological and environmental matrices.
Formal Definitions and Conceptual Framework
The scientific community has established precise definitions for EDCs based on their capacity to interfere with hormonal systems. In 2002, the World Health Organization (WHO) and the International Programme on Chemical Safety (IPCS) formally defined an EDC as "an exogenous substance or mixture that alters function(s) of the endocrine system and consequently causes adverse health effects in an intact organism, or its progeny or (sub-)populations" [6; 7; 8; 9; 10]. This definition emphasizes three critical elements: the exogenous origin of these compounds, their capacity to alter endocrine function, and the requirement that such alterations produce demonstrable adverse effects.
The Endocrine Society provides a complementary definition, characterising EDCs as "exogenous chemical[s], or mixture[s] of chemicals, that interfere with any aspect of hormone action" [8; 11; 12]. Synthesizing these definitions, EDCs can be understood as substances, whether natural or synthetic, that disrupt the production, distribution, function, metabolism, or excretion of hormones in biological systems [2; 13].
An important conceptual distinction must be drawn between EDCs and endocrine modulators. Endocrine modulators are compounds that interact with hormonal systems without necessarily producing harmful disruption. Common dietary substances such as coffee, chocolate, and soy products contain compounds that may modulate endocrine function, though their impact is typically dose-dependent and not inherently pathological [14]. In contrast, EDCs are characterised by their capacity to produce adverse health effects through endocrine disruption, distinguishing them from benign modulators.
Since the early 2000s, recognition of EDCs as a global public health issue has steadily increased, driven by evidence of their biological activity even at low environmental doses and their persistence in both environmental and biological matrices [1]. Exposure to EDCs is now understood to be a worldwide phenomenon affecting current and future human health across generations.
Classification and Major Sources of EDCs
EDCs constitute a remarkably heterogeneous group, derived from industrial processes, agricultural applications, consumer goods, medical products, and even natural sources [10; 14; 15; 16]. Due to their extensive production and ubiquitous use in modern society, human exposure to EDCs is widespread and, for many individuals, virtually unavoidable [3; 11; 17]. Figure 1 shows sources from which EDCs are derived.
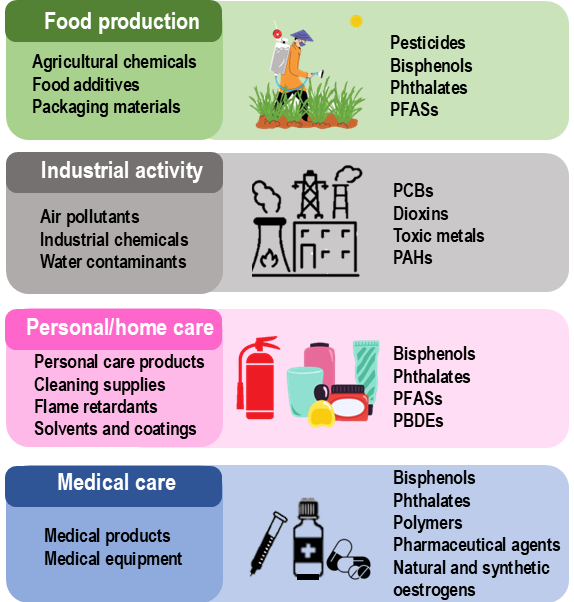
Plastics and Plasticizers
Bisphenol A (BPA) and Related Compounds: BPA represents one of the most extensively investigated EDCs. As a high-production volume chemical, BPA is used primarily in the manufacture of polycarbonate plastics and epoxy resins [4; 10]. These materials are ubiquitous in modern life, appearing in food and beverage packaging, canned food linings, water supply infrastructure, medical devices, thermal receipts, and children's toys [1; 4; 5; 7; 10; 12]. Primary exposure routes include dietary intake (the dominant pathway), dermal absorption, and inhalation [4; 10]. Examples of exposure sources of bisphenol and/or alternatives used in plastics [26]:
Lifestyle factors, e. g.,
• Diet
• Use of plastic products (leaching)
• Use of personal care products
• Household items
• Clothing
Environmental factors such as indoor/outdoor air/dust
Trans-placental transfer
Biomedical procedures, e. g.,
• Dental treatments
• Dialysis and infusions
• Medical implants
Residential location, e.g.,
• Fence line communities near plastic production /recycling facilities or landfills
• Industrial area
• Area of air/water/food contamination
Occupation, e.g.,
• Plastic production
• Plastic recycling (formal and informal)
• Waste management
• Non-plastic related such as cashiers
Following regulatory restrictions on BPA use in certain applications, manufacturers introduced substitute compounds such as Bisphenol S (BPS) and Bisphenol F (BPF). However, emerging evidence suggests that these substitutes may exhibit similar or potentially greater endocrine-disrupting properties compared to the original compound they were designed to replace, raising concerns about regrettable substitution [1; 8; 11].
Phthalates (PAEs): Phthalates represent the most widely used plasticizers globally, with annual consumption exceeding 7.5 million tons. Phthalates represent the most widely used plasticizers globally, with annual consumption exceeding 7.5 million tons [8]. These compounds are added to plastics to increase flexibility and durability, and they are found in diverse products including toys, medical devices (such as tubing and blood storage bags), cosmetics, personal care products, vinyl flooring, adhesives, detergents, and food packaging materials [5; 7; 8; 10; 12; 18].
Per- and Polyfluoroalkyl Substances (PFAS)
PFAS constitute a large family of man-made chemicals, comprising over 4,700 different substances characterized by their exceptional chemical stability. These compounds possess unique properties including oil and water repellence and thermal stability, making them valuable for numerous industrial and consumer applications [19; 20]. Common sources of PFAS exposure include firefighting foams, non-stick cookware, food packaging materials, textiles, and electronics manufacturing [1; 5; 7; 10; 19].
PFAS are notable for their environmental persistence and bioaccumulation potential, properties conferred by the exceptionally strong carbon-fluorine bond that characterizes these molecules [10; 19; 20; 21]. Perfluorooctanoic acid (PFOA) and perfluorooctanesulfonic acid (PFOS) represent the two most extensively studied and widely distributed PFAS compounds [19; 20].
Industrial and Agricultural Pollutants
Polychlorinated Biphenyls (PCBs): Historically, PCBs were used extensively in commercial and industrial applications. They were employed in electrical equipment (as insulating fluids in transformers and capacitors), as plasticizers, pigments, and flame retardants, and as cooling agents in electronic transformers and capacitors [7; 10; 22].
PCBs were banned in 1979 due to their high toxicity and perceived threats to human health and wildlife [23]. Despite this ban, PCB residues remain in the environment and can still be found in products such as electrical equipment, oil, cable, insulation, plastics, and paint due to their persistence and bioaccumulation [5; 10; 23].
PCBs are classified as persistent organic pollutants (POPs) with significant bioaccumulation potential [7; 10; 22].
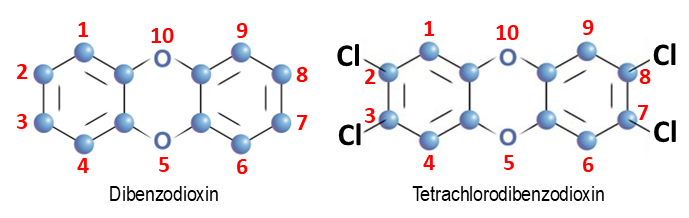
Dioxins: Dioxins are highly toxic compounds primarily generated as industrial byproducts, especially during waste incineration and pesticide manufacturing processes [14; 17; 22]. The term “dioxins” refers to a family of structurally related chemicals characterized by a core framework known as the dibenzo‑p‑dioxin skeleton, comprising two benzene rings connected by two oxygen bridges. This structure can accommodate various substituent atoms, with chlorine being the most prevalent, resulting in a class of compounds known as polychlorinated dibenzo‑p‑dioxins (PCDDs) [22]. Among these, TCDD (2,3,7,8‑tetrachlorodibenzo‑p‑dioxin) is recognized as the most toxic, distinguished by four chlorine atoms positioned at the 2, 3, 7, and 8 locations on the aromatic rings (Figure 1).
Pesticides: Pesticides are chemical substances deliberately formulated to control unwanted organisms, including insects (insecticides), weeds (herbicides), and fungi (fungicides). Rather than representing a single compound, they comprise a diverse family of synthetic mixtures that combine active ingredients with stabilisers, solvents, and dispersants to enhance field performance. Their extensive use in modern agriculture has made human exposure virtually unavoidable. Residues may persist on fruits and vegetables, leach into groundwater, drift through the air during spraying, and accumulate in household dust. Well‑studied examples include organochlorines such as DDT and its breakdown product DDE, herbicides like atrazine, fungicides such as vinclozolin, and organophosphates including chlorpyrifos and malathion [5; 7; 15; 23; 24; 25]. Organochlorine pesticides (OCPs), a class of synthetic chlorinated hydrocarbons, were widely applied from the 1940s through the 1960s. Many were subsequently banned in the United States due to their high toxicity, persistence, lipid solubility, bioaccumulation, and long environmental half‑life of 10–15 years. Each of these compounds has been linked to reproductive harm, developmental delays, or thyroid disruption, underscoring the dual identity of pesticides as both crop protectors and endocrine disruptors [10; 14; 25].
Flame Retardants: Flame retardants are a diverse category of chemicals that are widely used in commercial and household products due to their resistance to flammability. This category includes the legacy polybrominated diphenyl ethers (PBDEs) and organophosphorus flame retardants (OPFRs), which are incorporated into furniture foam, electronics, and textiles to meet fire safety standards [1; 7; 15; 16; 23; 25]. The legacy brominated compounds historically used in large volumes, many of which have since been banned or phased out due to environmental and health concerns, but they persist in the environment and household dust [23].
Additional Significant Sources
Heavy Metals: Certain toxic metals, including cadmium, mercury, and lead, exhibit endocrine-disrupting properties [1; 5; 22]. Cadmium is often released through industrial processes and cigarette smoke. Mercury is commonly introduced into ecosystems via coal combustion and artisanal gold mining. Lead was historically used in paints, gasoline, and plumbing. These metals are persistent in the environment and bioaccumulate in human tissues, making them a significant global health concern.
Natural and Synthetic Oestrogens: Environmental oestrogens originate from both agricultural sources (livestock excretion) and human sources (urinary excretion of pharmaceutical compounds). Notable examples include the synthetic oestrogen diethylstilbestrol (DES), historically prescribed to prevent miscarriage, and natural phytoestrogens such as genistein and daidzein, which are plant-derived compounds found in soy products and legumes [4; 10; 14;15].
Discussion
Endocrine Disrupting Chemicals (EDCs) represent a class of environmental contaminants whose significance lies not only in their ability to interfere with hormonal function but in their persistence and ubiquity across ecosystems. Leading health authorities stress that the defining characteristic of an EDC is the demonstrable harm it produces, yet what makes these chemicals particularly insidious is their integration into the global environment and their resistance to degradation.
The heterogeneity of EDCs underscores their pervasive presence in modern life. They are embedded in plastics, food packaging, pesticides, electronics, and personal care products, ensuring that exposure is continuous and unavoidable. High‑production volume chemicals such as bisphenols and phthalates exemplify this near‑constant contact, while the environmental persistence of PFAS, PCBs, and certain pesticides means they remain active for decades, accumulating in soils, waterways, and human tissues. This persistence magnifies their impact, creating long‑term cycles of exposure that extend across generations.
Compounding the challenge is the phenomenon of “regrettable substitution,” where banned EDCs are replaced with structurally similar alternatives that prove equally harmful. This cycle perpetuates environmental contamination and highlights the difficulty of achieving meaningful regulatory progress. Ultimately, the defining feature of EDCs is not only their endocrine activity but their enduring presence in the environment, which ensures that their health effects are widespread, cumulative, and deeply embedded in the fabric of everyday life.
Conclusion
In light of this ubiquity and diversity, it is evident that human exposure to EDCs is a widespread and virtually unavoidable feature of contemporary existence. This reality positions EDCs not merely as a toxicological issue, but as a fundamental and urgent public health priority. Addressing the risks posed by this complex group of chemicals requires a concerted effort towards developing stricter regulatory frameworks, promoting safer alternatives, and enhancing public awareness to mitigate exposure and safeguard health for current and future generations.
References
- Andonotopo, W., Bachnas, M. A., Dewantiningrum, J., Pramono, M. B. A., Akbar, M. I. A., Darmawan, E., Sanjaya, N. H., Aldiansyah, D., Sulistyowati, S., Stanojevic, M., & Kurjak, A. (2025). Endocrine disrupting chemicals: Translating mechanisms into perinatal risk assessment. Journal of Perinatal Medicine. Advance online publication. https://doi.org/10.1515/jpm-2025-0259
- Cortes-Ramirez, S. A., Ho, S.-M., & Leung, Y.-K. (2024). Endocrine-disrupting chemicals: A looming threat to current and future generations. International Journal of Molecular Sciences, 25(15), 8222. https://doi.org/10.3390/ijms2515822
- Derakhshan, A., Shu, H., Broeren, M. A. C., Kortenkamp, A., Lindh, C. H., Demeneix, B., Peeters, R. P., Bornehag, C.-G., & Korevaar, T. I. M. (2023). Association of endocrine disrupting chemicals exposure with human chorionic gonadotropin concentrations in pregnancy. Environment International, 178, 108091. https://doi.org/10.1016/j.envint.2023.108091
- Gonsioroski, A., Mourikes, V. E., & Flaws, J. A. (2020). Endocrine disruptors in water and their effects on the reproductive system. International Journal of Molecular Sciences, 21(6), 1929. https://doi.org/10.3390/ijms21061929
- Ma, C.-X., Ma, X.-N., Li, H.-L., Mauricio, D., & Fu, S.-B. (2025). Endocrine-disrupting chemicals exposure: Cardiometabolic health risk in humans. Cardiovascular Diabetology, 24, 381. https://doi.org/10.1186/s12933-025-02938-8
- Bonato, M., Corrà, F., Bellio, M., Guidolin, L., Tallandini, L., Irato, P., & Santovito, G. (2020). PFAS environmental pollution and antioxidant responses: An overview of the impact on human field. International Journal of Environmental Research and Public Health, 17(21), 8020. https://doi.org/10.3390/ijerph17218020
- Cargnelutti, F., Di Nisio, A., Pallotti, F., Sabovic, I., Spaziani, M., Tarsitano, M. G., Paoli, D., & Foresta, C. (2021). Effects of endocrine disruptors on fetal testis development, male puberty, and transition age. Endocrine, 72, 358–374. https://doi.org/10.1007/s12020-020-02436-9
- Haverinen, E., Fernandez, M. F., Mustieles, V., & Tolonen, H. (2021). Metabolic syndrome and endocrine disrupting chemicals: An overview of exposure and health effects. International Journal of Environmental Research and Public Health, 18(24), 13047. https://doi.org/10.3390/ijerph182413047
- Mokra, K. (2021). Endocrine disruptor potential of short- and long-chain perfluoroalkyl substances (PFASs)—A synthesis of current knowledge with proposal of molecular mechanism. International Journal of Molecular Sciences, 22(4), 2148. https://doi.org/10.3390/ijms22042148
- Di Pietro, G., Forcucci, F., & Chiarelli, F. (2023). Endocrine disruptor chemicals and children’s health. International Journal of Molecular Sciences, 24(3), 2671. https://doi.org/10.3390/ijms24032671
- Land, K. L., Miller, F. G., Fugate, A. C., & Hannon, P. R. (2022). The effects of endocrine-disrupting chemicals on ovarian- and ovulation-related fertility outcomes. Molecular Reproduction and Development, 89(6), 608–631. https://doi.org/10.1002/mrd.23652
- Trasande, L., & Sargis, R. M. (2024). Endocrine-disrupting chemicals: Mainstream recognition of health effects and implications for the practicing internist. Journal of Internal Medicine, 295(2), 259–274. https://doi.org/10.1111/joim.13748
- Leung, Y.-K. (2023). A silent threat: Exploring the impact of endocrine disruption on human health. International Journal of Molecular Sciences, 24(12), 9790. https://doi.org/10.3390/ijms24129790
- Guarnotta, V., Amodei, R., Frasca, F., Aversa, A., & Giordano, C. (2022). Impact of chemical endocrine disruptors and hormone modulators on the endocrine system. International Journal of Molecular Sciences, 23(10), 5710. https://doi.org/10.3390/ijms23105710
- Hall, J. M., & Greco, C. W. (2020). Perturbation of nuclear hormone receptors by endocrine disrupting chemicals: Mechanisms and pathological consequences of exposure. Cells, 9(1), 13. https://doi.org/10.3390/cells9010013
- Shulhai, A.-M., Palanza, P., & Street, M. E. (2024). Current evidence on the effects of endocrine-disrupting chemicals (EDCs) on bone growth and health. Exposure and Health, 16, 1001–1025. https://doi.org/10.1007/s12403-023-00607-3
- Onyebuchi, C., Muogbo, I., Chukwurah, C. O., Abdulkadir, M., Gordon, O. P., Ogundare, S. A., & Agaba, I. (2025). [Article title not provided]. medtigo Journal of Pharmacology, 2(3). https://doi.org/10.63096/medtigo30612310
- Rickard, B. P., Rizvi, I., & Fenton, S. E. (2022). Per- and poly-fluoroalkyl substances (PFAS) and female reproductive outcomes: PFAS elimination, endocrine-mediated effects, and disease. Toxicology, 465, 153031. https://doi.org/10.1016/j.tox.2021.153031
- Di Nisio, A., Lopez-Espinosa, M.-J., & Foresta, C. (2022). Editorial: Emerging chemical risks for human health: Endocrine disruption by per- and poly-fluorinated alkyl substances (PFAS). Frontiers in Endocrinology, 12, Article 813785. https://doi.org/10.3389/fendo.2021.813785
- Panieri, E., Buha Đorđevic, A., & Saso, L. (2021). Endocrine disruption by PFAS: A major concern associated with legacy and replacement substances. Arhiv za farmaciju, 71, 429–454. https://doi.org/10.5937/arhfarm71-34197
- Pezeshki, H., Rajabi, S., Hashemi, M., Moradalizadeh, S., & Nasab, H. (2025). Per- and poly-fluoroalkyl substances as forever chemicals in drinking water: Unraveling the nexus with obesity and endocrine disruption – A mini review. Heliyon, 11, e42782. https://doi.org/10.1016/j.heliyon.2025.e42782
- Zhang, X., Flaws, J. A., Spinella, M. J., & Irudayaraj, J. (2023). The relationship between typical environmental endocrine disruptors and kidney disease. Toxics, 11(32). https://doi.org/10.3390/toxics11010032
- Alsen, M., Sinclair, C., Cooke, P., Ziadkhanpour, K., Genden, E., & van Gerwen, M. (2021). Endocrine disrupting chemicals and thyroid cancer: An overview. Toxics, 9(1), 14. https://doi.org/10.3390/toxics9010014
- Birru, R. L., Liang, H.-W., Farooq, F., Bedi, M., Feghali, M., Haggerty, C. L., Mendez, D. D., Catov, J. M., Ng, C. A., & Adibi, J. J. (2021). A pathway level analysis of PFAS exposure and risk of gestational diabetes mellitus. Environmental Health, 20(63). https://doi.org/10.1186/s12940-021-00740-z
- Iwobi, N., & Sparks, N. R. (2023). Endocrine disruptor-induced bone damage due to hormone dysregulation: A review. International Journal of Molecular Sciences, 24(8263). https://doi.org/10.3390/ijms24098263
- Seewoo, B. J., Wong, E. V. S., Mulders, Y. R., Gozt, A., Elagali, A., Symeonides, C., & Dunlop, S. A. (2024). A systematic evidence map protocol for mapping global exposure to bisphenols and their alternatives and social and environmental justice implications. Environment International, 194, 109091. https://doi.org/10.1016/j.envint.2024.109091
