PFAS: The “Forever Chemicals” Shaping Our Health

Table of Contents
Abstract
Per- and polyfluoroalkyl substances (PFAS), commonly known as "forever chemicals," represent a class of thousands of synthetic compounds characterised by exceptional environmental persistence and widespread human exposure. Engineered since the 1940s for their ability to repel water, oil, and heat, PFAS have become ubiquitous in consumer products and environmental matrices. Their molecular stability, conferred by strong carbon-fluorine bonds, renders them virtually indestructible, leading to bioaccumulation in human tissues with elimination half-lives measured in years.
Human exposure occurs through multiple pathways: contaminated drinking water, dietary sources including fish and seafood that bioaccumulate PFAS, consumer products such as nonstick cookware and food packaging, and indoor environments where PFAS migrate from treated furnishings into dust and air. Occupational exposure in firefighting, chemical manufacturing, and related industries results in serum concentrations orders of magnitude higher than background levels.
PFAS function as endocrine-disrupting chemicals that mimic endogenous fatty acids and hormones. Through competitive binding to nuclear receptors, including peroxisome proliferator-activated receptors, androgen receptors, oestrogen receptors, and thyroid hormone receptors, PFAS disrupt fundamental regulatory pathways controlling metabolism, reproduction, immune function, and development. The International Agency for Research on Cancer has classified PFOA as carcinogenic to humans (Group 1), with strong associations documented for renal and testicular cancers. Additional health effects include thyroid dysfunction, metabolic syndrome, obesity, cardiovascular disease, reduced fertility, adverse birth outcomes, immunosuppression with reduced vaccine efficacy, and neurodevelopmental impacts including cognitive decline and ADHD. Critically, PFAS cross the placental barrier and concentrate in breast milk, with evidence suggesting potential transgenerational epigenetic effects.
While individual exposure reduction strategies offer meaningful protection, comprehensive solutions require regulatory reform, class-based chemical management, investment in remediation technologies, and precautionary evaluation of persistent synthetic chemicals. The PFAS crisis underscores the urgent need for protective policy interventions to safeguard current and future generations.
Keywords: PFAS, forever chemicals, endocrine disruptors, bioaccumulation, nuclear receptors, carcinogenicity, developmental toxicity, drinking water contamination
Introduction
Per- and polyfluoroalkyl substances (PFAS) represent a vast and complex family of synthetic organofluorine compounds that have seen global industrial application since the late 1940s and 1950s [1; 2; 3; 4]. Figure 1 shows Timeline of PFAS development from 1938 till present.
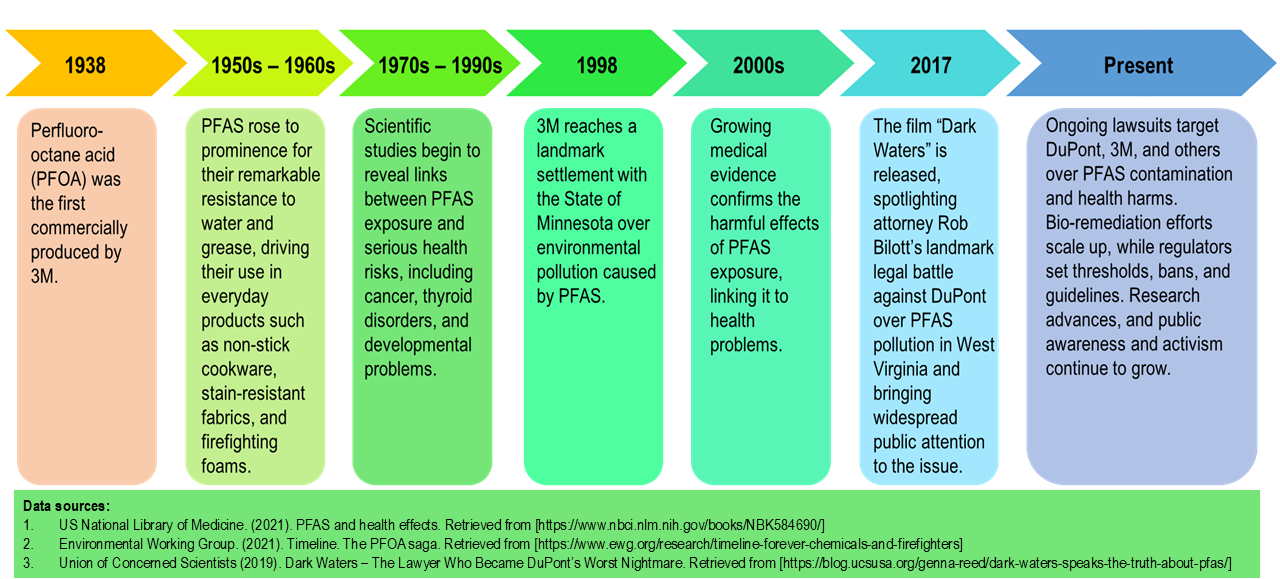
Figure 1 Timeline of PFAS development from 1938 till present (adapted from Lee et al. 2025)
Often characterised as "forever chemicals," PFAS are defined by their extraordinary resistance to biological, chemical, and thermal degradation. This extreme stability allows them to persist indefinitely within environmental matrices and bioaccumulate across the trophic levels of living organisms [3; 5; 6; 7].
While current chemical registries, such as the EPA’s CompTox and the OECD database, identify between 9,000 and 15,000 distinct structures, the broader universe of PFAS molecules may theoretically exceed millions [7; 8; 9; 10]. Despite this diversity, all PFAS share a foundational chemical signature: an aliphatic carbon chain where hydrogen atoms have been either fully or partially replaced by fluorine [8; 9].
Acronyms Explained
The PFAS family encompasses a wide range of acronyms. Table 1 provides definitions of several key terms.
Table 1 PFAS acronyms explained
Acronym | Full Name | Notes |
PFAS | Per- and polyfluoroalkyl substances | Large family of synthetic chemicals, often called “forever chemicals.” |
PFOA | Perfluorooctanoic acid | Once widely used in Teflon™ and stain-resistant products; linked to cancer and immune disorders. |
PFOS | Perfluorooctanesulfonic acid | Common in firefighting foams, stain-resistant fabrics, and food packaging; highly persistent and toxic. |
PFAA | Perfluoroalkyl acids | Broad subgroup of PFAS that includes carboxylic acids (like PFOA, PFBA) and sulfonic acids (like PFOS, PFBS); often used as reference category in toxicological studies. |
PFBA | Perfluorobutanoic acid | A short-chain PFAA; less bioaccumulative than long-chain PFAS but more mobile in water and harder to remove with standard filters. |
PFBS | Perfluorobutanesulfonic acid | Short-chain replacement for PFOS in some industrial uses; considered less bioaccumulative but still persistent and environmentally concerning. |
PFNA | Perfluorononanoic acid | A nine-carbon perfluoroalkyl carboxylic acid; highly bioaccumulative and associated with developmental and metabolic effects. |
PFDA | Perfluorodecanoic acid | A ten-carbon perfluoroalkyl carboxylic acid; long-chain PFAS with strong protein-binding affinity and endocrine-disrupting potential. |
NON | Perfluorononanoic acid (PFNA) | Alternate label used in toxicology studies; binds to androgen receptors and contributes to reproductive toxicity. |
HEP | Perfluoroheptanoic acid (PFHpA) | A seven-carbon perfluoroalkyl carboxylic acid; more mobile in water, less bioaccumulative, but still environmentally persistent. |
NNN | Perfluorononane sulfonic acid (PFNS) | A nine-carbon perfluoroalkyl sulfonic acid; structurally related to PFOS; interacts with androgen receptors and raises concern for endocrine disruption. |
Chemistry of PFAS
The aliphatic nature of the PFAS backbone is central to its toxicological profile. Unlike aromatic compounds, which consist of stable carbon rings, the aliphatic backbone of PFAS is comprised of straight or branched chains of carbon atoms [8; 9]. In these structures, the substitution of hydrogen with fluorine creates the carbon-fluorine (C–F) bond, which is recognised as the strongest and shortest single covalent bond in organic chemistry.
Because fluorine is the most electronegative element, it effectively "shields" the carbon backbone (Figure 2). This creates a helical sheath of fluorine atoms that renders the entire aliphatic chain nearly impervious to heat, water, and metabolic breakdown [2; 3; 11].
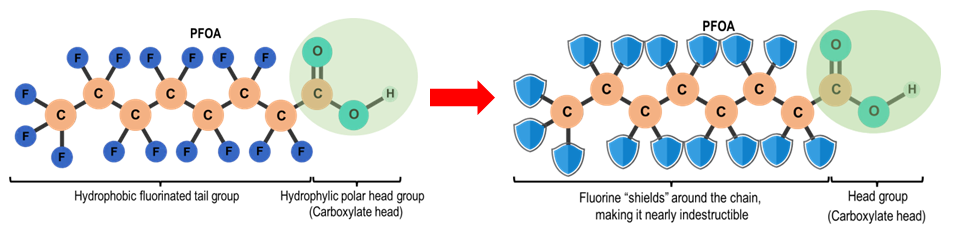
Figure 2 Chemical structure of PFAS (PFOA) head and tail
- Molecular Architecture
The unique performance and environmental persistence of PFAS are direct results of their specific molecular architecture. They typically employ an amphiphilic (or amphipathic) design consisting of two primary components: a tail and a head.
A Hydrophobic Tail: This component consists of an aliphatic carbon chain where the C–H bonds have been substituted with C–F bonds [4; 6; 12]. This tail is not only water-repellent (hydrophobic) but also oil-repellent (lipophobic/oleophobic). This rare combination makes PFAS invaluable for stain-resistant and grease-proof applications [3; 5; 13].
A Hydrophilic Head: Attached to the tail is a polar functional group that enables interaction with water and organic surfaces [6; 13; 11]. Common head groups include [14].
- Carboxylates (–COO⁻)
- Sulfonates (–SO₃⁻)
- Alcohols and Sulfonamides
- Carbon-Fluorine (C–F) Bond
PFAS are extremely stable. The core of their stability lies in the C–F bond [2; 3; 13; 15; 16]. Because of the high electronegativity of fluorine, the bond is exceptionally polarised and shielded, rendering it nearly impervious to hydrolysis, photolysis, and microbial metabolic processes. This makes PFAS effectively inert and resistant to high heat, strong acids, bases, and oxidising agents that prevents environmental degradation, cementing its status as a persistent "forever chemical" in the human body and the global ecosystem [3; 17; 18].
Perfluorooctanoic acid (PFOA), and example of PFAS), is built on an eight-carbon aliphatic chain that is fully fluorinated. This specific molecular architecture provides the high surface tension and chemical inertness required for industrial coatings.
Definitions and Classifications
The vast universe of PFAS is categorised based on the degree of fluorination, the length of the aliphatic carbon chain, and the molecular complexity (polymers vs. non-polymers).
- Perfluoroalkyl vs. polyfluoroalkyl
The distinction between these two groups depends on how many hydrogen atoms have been replaced by fluorine along the carbon backbone.
- Perfluoroalkyl substances: In these molecules, every hydrogen atom attached to the carbon backbone (except those within the functional head group) has been replaced by fluorine [2; 3; 17]. This fluorination results in a fully "shielded" chain with maximum stability.
- Polyfluoroalkyl substances: In these molecules, at least one, but not all, of the carbon-hydrogen bonds have been replaced by fluorine [2; 14; 17]. These are often referred to as "precursors" because the non-fluorinated parts of the chain are susceptible to degradation, eventually breaking down into terminal perfluoroalkyl acids.
- Chain Length: Long-chain vs. short-chain
Chain length significantly dictates how a PFAS molecule moves through the environment and how long the molecules stay in the human body (Table 2).
- Long-chain PFAS: Historically the most widely used PFAS. These include PFOA (8 carbons) and PFOS (8 carbons). Specifically, long-chain refers to perfluoroalkyl carboxylic acids with > 8 carbons and perfluoroalkane sulfonates > 6 carbons [3; 8;15]. They are highly bioaccumulative and have long biological half-lives.
- Short-chain PFAS: Compounds such as PFBS and PFBA have fewer carbon atoms. They were introduced as "safer" alternatives because they are less likely to bioaccumulate in fatty tissues. However, their smaller size makes them highly mobile in water and soil, allowing them to travel further from contamination sites and making them notoriously difficult to filter out of drinking water [3; 11; 15].
Table 2 Comparison of PFAS chain lengths
Feature | Long-Chain PFAS (e.g., PFOA, PFOS) | Short-Chain PFAS (e.g., PFBA, PFBS) |
Carbon count | > 8 (Carboxylates); > 6 (Sulfonates) | < 8 (Carboxylates; < 6 (Sulfonates) |
Bioaccumulation | High; binds to proteins and builds up in tissue | Lower; excreted more rapidly by the kidneys |
Environmental mobility | Lower; tends to stick to soil and sediment | Very High; moves rapidly through groundwater |
Filtration | Easier to remove with standard granular activated carbon (GAC) filters | Difficult; requires advanced Reverse Osmosis |
Regulatory status | Heavily restricted or banned globally | Often used as "replacement" chemicals |
- Polymers vs. non-polymers
The PFAS family is broadly divided by molecular weight and structural complexity:
- Non-Polymers: These include highly mobile perfluoroalkyl acids (PFAAs), such as PFOA and PFOS. They are the commonly detected in human blood and environmental samples.
- Polymers: This group includes high-molecular-weight substances, such as fluoropolymers (e.g., PTFE/Teflon), side-chain fluorinated polymers, and perfluoropolyethers [2; 12; 18]. While the polymers themselves are often large and less mobile, their manufacturing process often involves non-polymeric PFAS.
- PFAS precursors and terminal products
Many PFAS serve as precursors. These molecules can undergo partial transformation through biotic (microbial) or abiotic (chemical) processes in soil, air, or wastewater treatment plants [8; 9]. The result of this transformation is often a terminal, highly stable perfluoroalkyl acid (PFAA), such as PFOA or PFOS, which can no longer be broken down by natural processes [19; 20]. Figure 3 illustrates the transformation pathways of PFAS precursors.
Figure 3 PFAS precursor transformation pathways
Human Exposure Pathways
Human exposure to PFAS is characterised by its multi-modal nature, involving a complex interplay between contaminated environmental media and widespread commercial products. Because of their unique chemical stability, PFAS do not remain confined to their point of origin but migrate through ecosystems, eventually entering the human body through several direct and indirect routes.
These persistent chemicals reach us through an interconnected web of pathways that span our food, water, homes, and workplaces (Figure 4). Understanding how PFAS enter our bodies is the first step toward meaningful reduction of exposure.
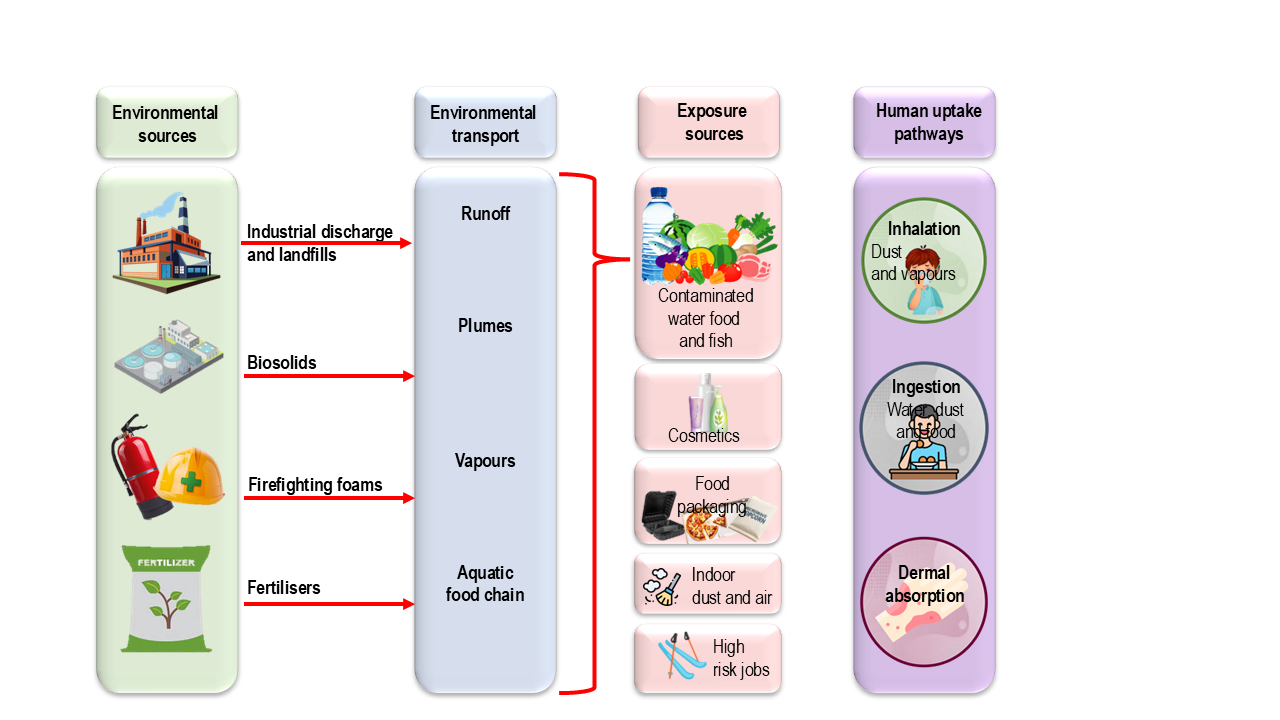
Figure 4 PFAS exposure sources and uptake pathways
- Ingestion: Dominant Pathway
For most individuals without occupational exposure, the ingestion of food and beverages represents the primary route of PFAS exposure [2, 3].
- Drinking Water: Contamination of public and private water systems is a global crisis. In the United States alone, it is estimated that over 200 million residents are served by water systems with detectable levels of PFAS [21]. These chemicals often enter the water table through industrial discharge, landfill leachate, and the use of firefighting foams.
- Dietary Sources: Food serves as a significant secondary source of ingestion. Aquatic ecosystems are particularly vulnerable; fish and seafood are known to bioaccumulate high concentrations of PFAS from contaminated water [2; 22]. Additionally, PFAS can enter the food chain via crops grown in soil treated with contaminated biosolids or irrigated with tainted water [23; 24].
- Indoor Environment and Dust Inhalation
Our homes, meant to be sanctuaries, can inadvertently become sites of chronic PFAS exposure. These chemicals do not stay locked in the products that contain them. Over time, PFAS migrate from treated materials into house dust and indoor air, creating a diffuse but persistent source of contamination throughout living spaces [3; 8; 25].
- Sources: Common contributors are often items chosen for their practical benefits: stain-resistant carpets that withstand spills, upholstered furniture treated to repel liquids, and water-repellent outdoor clothing brought indoors [3; 18; 26]. As these products age and degrade, they release PFAS particles that settle on surfaces and become suspended in the air we breathe.
- Paediatric vulnerability: Young children face disproportionate risk from this pathway. Their natural behaviours, crawling on floors, frequent hand-to-mouth contact, and playing close to the ground where dust accumulates, result in significantly higher exposure rates compared to adults in the same environment [8; 25; 27]. This is particularly concerning given children's vulnerable developmental stages and the potential for PFAS to interfere with growth and development.
- Consumer Product Contact
Beyond environmental contamination, PFAS are encounter through deliberate use of treated products. Direct exposure frequently occurs through daily interaction with household items. PFAS are used for their surfactant properties and resistance to heat and oil, leading to their inclusion in:
- Nonstick cookware: While stable at normal temperatures, PTFE-coated (non-stick) cookware can release PFAS precursors if overheated [26]. Particularly traditional Teflon-coated pans, can release PFAS compounds during cooking, especially when heated to high temperatures or when the coating becomes scratched or damaged [3; 4; 10;15; 26].
- Food contact materials: Grease-resistant treatments applied to microwave popcorn bags, fast-food wrappers, and pizza boxes allow PFAS to migrate directly into the foods they contact, particularly when heated [3; 4; 10;15; 26]. The convenience of these packaging materials comes with the hidden cost of chemical transfer from wrapper to meal.
- Personal care products: Cosmetics, certain brands of dental floss, and some sunscreens contain PFAS, either as intentional ingredients to improve texture and water resistance or as contaminants from manufacturing processes [3; 4; 10;15; 26]. These products are applied directly to skin or used in the mouth, facilitating absorption into the body.
- Occupational Exposure Risks
While the general population faces low to moderate chronic exposure, workers in certain professional sectors encounter PFAS at dramatically higher levels. Workers in these industries often exhibit serum PFAS concentrations that are several orders of magnitude higher than those of the general public [8; 11]. These sectors include:
- Chemical manufacturing: Workers involved in PFAS production or use face direct contact with concentrated forms of these substances.
- Aqueous film-forming foams (AFFF) Exposure: Firefighters represent perhaps the most studied high-exposure occupational group. Those who train with or deploy aqueous AFFF absorb PFAS through skin contact and inhalation [3; 8; 11]. Studies consistently show that firefighters' serum PFAS concentrations can reach levels several orders of magnitude, hundreds or even thousands of times, higher than those found in the public. This occupational exposure has been linked to increased rates of certain cancers and other health conditions among firefighters.
- Specialised industries: Professional ski waxers who work with fluorinated waxes inhale PFAS-laden particles regularly throughout their workday [3; 8; 11].
5. Cumulative effect
What makes PFAS exposure particularly insidious is its omnipresence. We are not exposed through one dramatic event, but rather through countless small encounters every day, a glass of water, a meal from a takeout container, time spent on a treated carpet, application of cosmetics. These exposures accumulate in our bodies over time, building up because PFAS are eliminated extremely slowly, with half-lives measured in years rather than days.
Recognising these pathways allows for targeted intervention. While we cannot eliminate all exposure in our current chemical landscape, understanding where PFAS come from empowers us to make informed choices about the water we drink, the products we bring into our homes, and the materials that contact our food.
Molecular Mechanisms of Endocrine Disruption
- PFAS as Molecular Infiltrators
PFAS are often described as “molecular infiltrators” because their chemical structure allows them to mimic endogenous substances such as fatty acids and hormones. This mimicry enables them to penetrate biological control centres and bind to the nuclear receptor superfamily [18; 28]. Once inside the cell, PFAS can be transported into the nucleus, where they act as analogues or antagonists, activating or blocking receptors that regulate growth, development, and metabolism [18; 28].
Mechanism of entry: The infiltration mechanism operates through structural mimicry. Many PFAS are structural homologs of fatty acids, sharing similar molecular architecture with natural lipids [18; 28]. This resemblance facilitates their absorption and transport to the nucleus [28]. For example, PFOA can be transported directly into the cell nucleus, where it functions as a structural analogue to activate the Peroxisome Proliferator-Activated Receptor alpha (PPARα) [28].
The disruption occurs through competitive binding: PFAS infiltrate the ligand-binding pockets of receptors, physically displacing natural hormones such as testosterone or thyroxine (T4) [27; 29]. This competitive displacement represents a critical mechanism through which PFAS exert their endocrine-disrupting effects, essentially hijacking cellular signalling pathways intended for endogenous hormones.
2. Nuclear Receptor Targets
PFAS interact with multiple members of the nuclear receptor superfamily, each interaction carrying distinct physiological consequences:
PPARs: PFAS bind to multiple PPAR isoforms. These include PPARα, PPARγ, and PPARβ/δ [19; 28]. These receptors serve as master regulators of lipid homeostasis, adipocyte differentiation, and glucose metabolism [18; 28]. Through PPAR activation, PFAS can induce the liver to increase fatty acid metabolism and promote adipogenesis, the development of new fat cells [18; 28]. This mechanism contributes to metabolic disruption and may play a role in obesity and metabolic syndrome.
Androgen receptor (AR): Emerging PFAS compounds, including NON (perfluorononanoic acid (PFNA)), HEP (perfluoroheptanoic acid (PFHpA)), and NNN (perfluorononane sulfonic acid (PFNS or PFOS‑related)), have demonstrated the ability to bind to the human androgen receptor [27]. These compounds act as competitive antagonists, occupying the receptor's binding site to prevent testosterone from exerting its normal physiological effects [27]. This antagonism can lead to reduced sperm quality and impaired reproductive function in males.
Oestrogen receptors: Certain PFAS, including PFOA, PFOS, PFNA, and PFDA, can activate ERα, the primary oestrogen receptor isoform [18; 28]. This pro-oestrogenic activity may stimulate the proliferation of hormone-dependent cancers, particularly breast cancer, by mimicking the growth-promoting effects of endogenous oestrogen.
Thyroid hormone receptors (TR): PFAS interfere with the hypothalamic-pituitary-thyroid axis by binding to thyroid hormone receptors [15; 30]. This disruption can impair neurodevelopment, particularly during critical windows of foetal and early childhood development, and alter basal metabolic rates throughout life.
Additional receptor targets: PFAS also interact with the Liver X Receptor (LXR), which regulates cholesterol homeostasis, and the Pregnane X Receptor (PXR), which plays a central role in detoxifying foreign substances [18; 20]. These interactions contribute to the broad metabolic effects observed with PFAS exposure.
- Downstream Biological Consequences
The molecular infiltration of nuclear receptors by PFAS triggers cascading effects on gene transcription and cellular function:
Steroidogenesis interference: By binding to nuclear receptors, PFAS alter the expression of critical enzymes in the steroid hormone synthesis pathway. This includes aromatase (CYP19A1), which converts androgens to oestrogens, and steroidogenic acute regulatory protein (StAR), which facilitates the rate-limiting step in steroid hormone production [13; 18; 32]. These disruptions compromise the production of sex hormones such as oestradiol and progesterone, contributing to reproductive and developmental abnormalities.
Epigenetic alterations: High-level PFAS exposure can induce DNA methylation changes and histone modifications [32; 33]. These epigenetic modifications alter gene expression patterns without changing the underlying DNA sequence itself. Critically, such changes can persist long-term and may even be transmitted across generations, raising the possibility of transgenerational health effects [32; 33]. This mechanism suggests that PFAS exposure in one generation could influence the health outcomes of subsequent generations, even in the absence of direct exposure.
- Conceptual Framework
To understand this mechanism conceptually, imagine the cell's nucleus as a high-security command centre where nuclear receptors act as the biological "keyholes" controlling operations. PFAS act as counterfeit keys. Because their molecular structure so closely resembles the authorised "keys" (natural hormones and fatty acids) they can slip into the lock. Some PFAS function as agonists, turning the lock to initiate unauthorised operations such as excessive fat production. Others function as antagonists, jamming the lock and preventing authentic keys from working at all. This molecular mimicry allows PFAS to commandeer fundamental cellular processes, disrupting the carefully regulated hormonal systems that govern metabolism, reproduction, and development.
Human Health Outcomes of PFAS exposure
The environmental persistence of PFAS leads to significant accumulation in human tissues, resulting in a plethora of adverse health outcomes. While most research has focused on a small subset of "legacy" compounds such as PFOA and PFOS, emerging research indicates that newer alternatives, such as GenX and F-53B, may exhibit similar or even higher toxic potential [3; 8; 13; 14].
The breadth of health effects associated with PFAS exposure reflects their ability to disrupt multiple biological systems simultaneously, creating a complex toxicological profile that continues to unfold as research advances. Their impact spans multiple physiological systems, ranging from oncogenesis to the disruption of complex metabolic and developmental cycles.
- Carcinogenicity
The carcinogenic potential of PFAS has gained increasing recognition from international health authorities. The International Agency for Research on Cancer (IARC) recently classified PFOA as "carcinogenic to humans" (Group 1) and PFOS as "possibly carcinogenic" (Group 2B) [3; 7; 11]. These classifications reflect decades of accumulating evidence linking PFAS exposure to cancer development.
Renal and testicular cancer: Strong epidemiological links exist between high PFAS exposure and increased risk of renal and testicular cancers [13; 28]. These associations have been particularly well-documented in occupationally exposed populations and communities with contaminated drinking water.
Other malignancies: Investigations have also reported associations between PFAS concentrations and prostate, liver, breast, and pancreatic cancers [3; 13; 28]. The mechanisms underlying PFAS carcinogenicity appear multifactorial, involving both direct genotoxic effects and indirect promotion of tumour development through chronic inflammation, oxidative stress, and endocrine disruption.
2. Endocrine and Metabolic Disruption
PFAS act as potent endocrine-disrupting chemicals (EDCs) that interfere with hormone synthesis, transport, and receptor binding. This endocrine disruption manifests across multiple physiological systems, with particularly pronounced effects on thyroid function, metabolic homeostasis, and hepatic health [4; 28].
Thyroid dysfunction: PFAS are recognised as thyroid disruptors, frequently associated with the onset of hypothyroidism, altered thyroid-stimulating hormone (TSH) levels, and thyroid follicular cell adenomas [15; 18; 24; 26]. The mechanisms involve both direct interference with thyroid hormone synthesis and disruption of thyroid hormone transport proteins in circulation. Given the critical role of thyroid hormones in regulating metabolism, growth, and neurodevelopment, these disruptions carry significant health implications across human lifespan.
Metabolic syndrome and obesity: PFAS are classified as "obesogens" because they can prompt stem cells to differentiate into adipocytes and alter lipid metabolism [1; 4; 18; 20]. Exposure is linked to dyslipidemia, characterised by elevated cholesterol and triglyceride levels, type 2 diabetes, and increased body weight [4; 15; 18; 28]. These metabolic effects occur through multiple pathways, including activation of PPARs, disruption of insulin signalling, and interference with leptin and adiponectin secretion. The obesogenic effects of PFAS are particularly concerning given the global rise in metabolic disease and the potential for developmental programming of obesity risk during critical windows of exposure.
Liver toxicity: The liver represents a primary site of PFAS accumulation due to its role in xenobiotic metabolism and its high affinity for binding PFAS. This accumulation can cause non-alcoholic fatty liver disease (NAFLD), liver lesions, and abnormal levels of liver enzymes such as alanine aminotransferase (ALT) and gamma-glutamyl transferase (GGT) [4; 13; 26; 28]. The hepatotoxic effects of PFAS appear to involve mitochondrial dysfunction, oxidative stress, and dysregulation of lipid metabolism pathways.
3. Reproductive and Developmental Outcomes
PFAS can cross the placental barrier and are excreted in breast milk, exposing infants during critical windows of development [6; 24; 30; 33; 34]. This developmental exposure is particularly concerning given the vulnerability of developing organ systems to endocrine disruption.
Infertility: Exposure is linked to reduced fecundity, manifesting as longer time to conception, and conditions such as endometriosis, polycystic ovarian syndrome (PCOS), and primary ovarian insufficiency [1; 13; 28; 32]. These reproductive effects occur through disruption of steroidogenesis, interference with follicular development, and alterations in the hypothalamic-pituitary-gonadal axis.
Male reproductive health: Studies report poor sperm quality, lower sperm counts, and reduced testosterone production in men exposed to elevated PFAS levels [3; 16; 18; 27; 28]. The anti-androgenic effects of certain PFAS compounds contribute to these outcomes by competitively blocking androgen receptors and disrupting testicular steroidogenesis.
Adverse birth outcomes: Prenatal exposure is consistently associated with low birth weight, foetal growth restriction, miscarriage, and pregnancy-induced hypertension, including preeclampsia [1; 4; 6; 13; 24; 28; 35]. These effects appear to be mediated through placental dysfunction, oxidative stress, and disruption of critical developmental signalling pathways. The dose-response relationships observed in epidemiological studies suggest that even relatively low levels of maternal PFAS exposure can compromise foetal development.
4. Immunotoxicity and Respiratory Health
The immune system is considered one of the most sensitive targets for PFAS toxicity [38]. This immunosuppressive effect has profound implications for disease resistance and vaccine efficacy.
Vaccine response: High PFAS exposure in children is associated with a reduced antibody response to vaccines, including tetanus and diphtheria, potentially decreasing overall vaccine effectiveness [1; 4; 10; 13;15; 18; 26; 28]. This immunosuppressive effect occurs through disruption of B-cell maturation and antibody production, raising concerns about the adequacy of vaccine-induced immunity in highly exposed populations.
Asthma: Exposure is linked to an increased risk of asthma morbidity and impaired lung function, particularly in children [1; 4; 28]. The mechanisms underlying these respiratory effects may involve both immunological alterations and direct effects on airway epithelial cells and smooth muscle.
5. Cardiovascular and Renal Effects
Cardiovascular disease: Long-term PFAS exposure is associated with an increased risk of hypertension, myocardial infarction, and cardiovascular mortality [3; 4; 13; 28; 31; 35; 36]. These cardiovascular effects appear to be mediated through multiple pathways, including dyslipidemia, endothelial dysfunction, oxidative stress, and chronic low-grade inflammation. The relationship between PFAS exposure and cardiovascular disease represents a significant public health concern given the high prevalence of cardiovascular mortality globally.
Renal health: PFAS exposure is linked to chronic kidney disease, renal dysfunction, and reduced estimated glomerular filtration rates (eGFR) [1; 3; 4; 15; 28]. The kidneys play a dual role in PFAS toxicology: they are both a primary route of PFAS excretion and a target organ for PFAS-induced damage. The nephrotoxic effects may involve direct tubular toxicity, glomerular damage, and interference with renal transport proteins.
6. Neurodevelopmental and Mental Health
Cognitive impacts: Childhood exposure is linked to attention-deficit/hyperactivity disorder (ADHD), lower intelligence quotient (IQ) scores, and cognitive decline [1; 3; 16; 24]. These neurodevelopmental effects are particularly concerning because they occur during critical windows of brain development and may have lifelong consequences for educational achievement and cognitive function.
Mental well-being: Community studies highlight significant psychosocial stress, characterized by anxiety and depression due to the uncertainty of long-term health risks and living in contaminated environments [1; 11]. This psychological burden represents an often-overlooked dimension of PFAS toxicity, reflecting the broader social and emotional costs of environmental contamination.
Neurodegenerative risk: Emerging evidence suggests potential associations with the risk of Alzheimer's and Parkinson's diseases [11; 16; 28; 37]. While this research is still developing, the mechanisms may involve neuroinflammation, oxidative stress, and disruption of neurotransmitter systems. Given the aging global population, the potential contribution of environmental chemicals to neurodegenerative disease represents a critical area for future investigation.
The persistence of PFAS in the environment and bioaccumulation in human tissues have been linked to carcinogenic, endocrine, reproductive, immunological, cardiovascular, renal, and neurodevelopmental consequences. To provide a clear overview, Table 3 synthesizes key health outcomes associated with PFAS exposure, highlighting the mechanisms involved and the supporting evidence from epidemiological and experimental studies.
Table 3 Summary of major health outcomes associated with PFAS exposure, including affected systems, mechanisms of toxicity, and supporting references
Category | Health Outcomes | Notes & Mechanisms |
Carcinogenicity | - Renal & testicular cancer | IARC: PFOA = Group 1 carcinogen; PFOS = Group 2B possible carcinogen. Mechanisms include genotoxicity, chronic inflammation, oxidative stress, and endocrine disruption. |
Endocrine & Metabolic Disruption | - Thyroid dysfunction (hypothyroidism, altered TSH, adenomas) | PFAS act as endocrine disruptors, interfering with hormone synthesis, transport, and receptor binding. Mechanisms include PPAR activation, insulin signaling disruption, mitochondrial dysfunction, and oxidative stress. |
Reproductive & Developmental Outcomes | - Infertility (endometriosis, PCOS, ovarian insufficiency) | PFAS cross placenta and are excreted in breast milk. Mechanisms include steroidogenesis disruption, anti-androgenic receptor binding, placental dysfunction, oxidative stress, and altered developmental signaling. |
Immunotoxicity & Respiratory Health | - Reduced vaccine response (tetanus, diphtheria) | PFAS suppress immune function by disrupting B-cell maturation and antibody production. Respiratory effects may involve immune alterations and direct airway toxicity. |
Cardiovascular & Renal Effects | - Cardiovascular disease (hypertension, myocardial infarction, mortality) | PFAS contribute via dyslipidemia, endothelial dysfunction, oxidative stress, and chronic inflammation. Kidneys are both excretion route and target organ, with tubular and glomerular toxicity. |
Neurodevelopmental & Mental Health | - Cognitive impacts (ADHD, lower IQ, cognitive decline) | PFAS exposure during brain development impairs cognition. Mechanisms include neuroinflammation, oxidative stress, neurotransmitter disruption. Psychosocial stress arises from contaminated environments and uncertainty of health risks. |
7. Conceptual Integration
Human exposure to PFAS can be conceptualized as installing faulty software into a biological computer. Once these "forever chemicals" enter the system, they embed themselves into the core operating code—proteins and receptors—of multiple biological "applications," including metabolism, immune function, and growth regulation. Because the body lacks an effective "uninstall" feature for these chemicals, they continue to run in the background for years, causing cumulative glitches that eventually manifest as system-wide failures such as cancer, metabolic disease, or reproductive dysfunction. This analogy captures both the persistent nature of PFAS and their ability to disrupt multiple interconnected biological systems simultaneously.
Conclusion: Confronting the PFAS Legacy
The story of PFAS represents one of the most complex environmental health challenges of our time. These synthetic molecules, engineered for their remarkable chemical stability, have become ubiquitous contaminants that persist in our water, air, soil, food, and bodies. Their designation as "forever chemicals" reflects both their environmental persistence and the enduring nature of the public health crisis they have created.
What emerges from the scientific literature is a portrait of chemicals that operate as molecular infiltrators, mimicking natural hormones and fatty acids to commandeer the biological machinery that controls metabolism, reproduction, immune function, and development. The consequences manifest across virtually every organ system; from cancer and cardiovascular disease to metabolic dysfunction, reproductive impairment, immunosuppression, and neurodevelopmental effects. PFAS do not simply cause isolated toxic effects but rather disrupt the integrated networks that maintain human health.
The epidemiological evidence has reached a threshold that demands action. The International Agency for Research on Cancer's classification of PFOA as carcinogenic to humans represents a watershed moment. Yet this addresses only one compound among thousands currently in commercial use. Many replacement compounds marketed as safer alternatives may exhibit comparable or greater toxicity, a pattern of "regrettable substitution" that highlights the inadequacy of compound-by-compound regulation.
Particularly troubling is the intergenerational dimension of PFAS toxicity. These chemicals cross the placental barrier and concentrate in breast milk, exposing vulnerable populations during critical developmental windows. Emerging evidence of epigenetic modifications suggests PFAS exposure may alter gene expression patterns across generations, potentially affecting individuals never directly exposed.
The crisis also illuminates pathways forward. At the individual level, informed choices about cookware, food packaging, water filtration, and consumer products can meaningfully reduce exposure. For families with young children, choosing untreated furnishings, frequent cleaning, and avoiding stain-resistant treatments can substantially lower exposure during vulnerable periods.
We cannot undo decades of PFAS production and environmental release, but we can determine how we respond to this legacy. The choices we make today, as individuals, communities, policymakers, and societies, will shape PFAS exposure for current and future generations. The science is clear: PFAS pose substantial risks to human health. The path forward requires translating this knowledge into protective action at every level, from kitchen choices to international chemical policy. In confronting the challenge of forever chemicals, we can forge a more thoughtful, precautionary approach to the synthetic substances we create and release into the world we inhabit.
Key Findings
Category | Key takeaway |
Chemical Architecture | PFAS are defined by a fully or partially fluorinated aliphatic carbon chain. The carbon-fluorine (C–F) bond is the strongest single covalent bond in organic chemistry, rendering these molecules effectively "indestructible" by natural processes. |
Bioaccumulation
| Their resistance to degradation allows them to persist indefinitely in the environment and accumulate in living organisms across all trophic levels. |
Molecular Mimicry | PFAS act as "molecular infiltrators" because their structure allows them to mimic natural fatty acids and hormones. |
Systemic Health Impacts | Exposure is linked to significant risks, including Group 1 carcinogenicity (PFOA), hypothyroidism, vaccine suppression, and metabolic syndrome. |
References
- Banwell, C., Housen, T., Smurthwaite, K., Trevenar, S., Walker, L., Todd, K., et al. (2021). Health and social concerns about living in three communities affected by per- and polyfluoroalkyl substances (PFAS): A qualitative study in Australia. PLoS ONE, 16(1), e0245141. https://doi.org/10.1371/journal.pone.0245141 (doi.org in Bing)
- Chaudhary, A., Bashir, W., Majid, A., Qasim, M., Bughio, E., Fatima, M., & Din, S. U. (2025). PFAS insights: A review of historical data, environmental applications, health effects, and pollution challenges in Pakistan. Environmental Science and Policy, 167, 104056. https://doi.org/10.1016/j.envsci.2025.104056
- Habib, Z., Song, M., Ikram, S., & Zahra, Z. (2024). Overview of per- and polyfluoroalkyl substances (PFAS), their applications, sources, and potential impacts on human health. Pollutants, 4(1), 136–152. https://doi.org/10.3390/pollutants4010009
- Pezeshki, H., Rajabi, S., Hashemi, M., Moradalizadeh, S., & Nasab, H. (2025). Pre- and poly-fluoroalkyl substances as forever chemicals in drinking water: Unravelling the nexus with obesity and endocrine disruption – A mini review. Heliyon, 11, e42782. https://doi.org/10.1016/heliyon.2025.e42782
- Birru, R. L., Liang, H.-W., Farooq, F., Bedi, M., Feghali, M., Haggerty, C. L., Mendez, D. D., Catov, J. M., Ng, C. A., & Adibi, J. J. (2021). A pathway level analysis of PFAS exposure and risk of gestational diabetes mellitus. Environmental Health, 20, 63. https://doi.org/10.1186/s12940-021-00740-z
- Jha, G., Kankarla, V., McLennon, E., Pal, S., Sihi, D., Dari, B., Diaz, D., & Nocco, M. (2021). Per- and polyfluoroalkyl substances (PFAS) in integrated crop–livestock systems: Environmental exposure and human health risks. International Journal of Environmental Research and Public Health, 18(23), 12550. https://doi.org/10.3390/ijerph182312550
- Sangkham, S., Phairuang, W., Pansakun, N., & Ford, A. T. (2025). Specific ambient PM2.5-bound perfluoroalkyl and polyfluoroalkyl substances (PFAS): A prominent human health risk issue that deserves attention? Environmental Science & Technology, 59(28), 14780–14783. https://doi.org/10.1021/acs.est.5c07694
- Cousins, I. T., DeWitt, J. C., Glüge, J., Goldenman, G., Herzke, D., Lohmann, R., Miller, M., Ng, C., Scheringer, M., Vierke, L., & Wang, Z. (2020). Strategies for grouping per- and polyfluoroalkyl substances (PFAS) to protect human and environmental health. Environmental Science: Processes & Impacts, 22(7), 1444–1460. https://doi.org/10.1039/d0em00147c
- De Silva, A. O., Armitage, J. M., Bruton, T. A., Dassuncao, C., Heiger-Bernays, W., Hu, X. C., Kärrman, A., Kelly, B., Ng, C., Robuck, A., Sun, M., Webster, T. F., & Sunderland, E. M. (2021). PFAS exposure pathways for humans and wildlife: A synthesis of current knowledge and key gaps in understanding. Environmental Toxicology and Chemistry, 40(3), 631–657. https://doi.org/10.1002/etc.4935
- Lee, J. C., Smaoui, S., Duffill, J., Marandi, B., & Varzakas, T. (2025). Research progress in current and emerging issues of PFASs’ global impact: Long-term health effects and governance of food systems. Foods, 14(6), 958. https://doi.org/10.3390/foods14060958
- Sukhram, S. D., Kim, J., Musovic, S., Anidugbe, A., Corte, E., Ahsan, T., Rofail, S., Mesquita, N., & Padilla, M. (2025). PFAS exposure, mental health, and environmental justice in the United States: Impacts on marginalized communities. International Journal of Environmental Research and Public Health, 22(7), 1116. https://doi.org/10.3390/ijerph22071116
- Mayilswami, S., Raval, N. P., Tomar, R., Sharma, S., Praveena, S. M., Kataria, N., Selvasembian, R., Shanmugam, S. R., Nath, R., Malakar, A., Dutta, S., & Mukherjee, S. (2025). Potential human health effects of per- and polyfluoroalkyl substances (PFAS) prevalent in aquatic environment: A review. Environmental Science: Advances, 4, 1939–1962. https://doi.org/10.1039/d4va00405a
- Perera, D. C., & Meegoda, J. N. (2024). PFAS: The journey from wonder chemicals to environmental nightmares and the search for solutions. Applied Sciences, 14(19), 8611. https://doi.org/10.3390/app14198611
- Manojkumar, Y., Pilli, S., Venkateswara Rao, P., & Tyagi, R. D. (2023). Sources, occurrence and toxic effects of emerging per- and polyfluoroalkyl substances (PFAS). Neurotoxicology and Teratology, 97, 107174. https://doi.org/10.1016/j.ntt.2023.107174
- Coperchini, F., Croce, L., Ricci, G., Magri, F., Rotondi, M., Imbriani, M., & Chiovato, L. (2021). Thyroid disrupting effects of old and new generation PFAS. Frontiers in Endocrinology, 11, 612320. https://doi.org/10.3389/fendo.2020.612320
- Kikanme, K. N., Dennis, N. M., Orikpete, O. F., & Ewim, D. R. E. (2024). PFAS in Nigeria: Identifying data gaps that hinder assessments of ecotoxicological and human health impacts. Heliyon, 10, e29922. https://doi.org/10.1016/j.heliyon.2024.e29922
- Gonsioroski, A., Mourikes, V. E., & Flaws, J. A. (2020). Endocrine disruptors in water and their effects on the reproductive system. International Journal of Molecular Sciences, 21(6), 1929. https://doi.org/10.3390/ijms21061929
- Mokra, K. (2021). Endocrine disruptor potential of short- and long-chain perfluoroalkyl substances (PFASs)—A synthesis of current knowledge with proposal of molecular mechanism. International Journal of Molecular Sciences, 22(4), 2148. https://doi.org/10.3390/ijms22042148
- Guelfo, J. L., Korzeniowski, S., Mills, M. A., Anderson, J., Anderson, R. H., Arblaster, J. A., Conder, J. M., Cousins, I. T., Dasu, K., Henry, B. J., Lee, L. S., Liu, J., McKenzie, E. R., & Willey, J. (2021). Environmental sources, chemistry, fate, and transport of per- and polyfluoroalkyl substances: State of the science, key knowledge gaps, and recommendations presented at the August 2019 SETAC Focus Topic Meeting. Environmental Toxicology and Chemistry, 40(12), 3234–3260. https://doi.org/10.1002/etc.5182
- Panieri, E., Buha Đorđevic, A., & Saso, L. (2021). Endocrine disruption by PFAS: A major concern associated with legacy and replacement substances. Arhiv za farmaciju, 71, 429–454. https://doi.org/10.5937/arhfarm71-34197
- Salvatore, D., Mok, K., Garrett, K. K., Poudrier, G., Brown, P., Birnbaum, L. S., Goldenman, G., Miller, M. F., Patton, S., Poehlein, M., Varshavsky, J., & Cordner, A. (2022). Presumptive contamination: A new approach to PFAS contamination based on likely sources. Environmental Science & Technology Letters, 9(11), 983–990. https://doi.org/10.1021/acs.estlett.2c00502
- Dimitrakopoulou, M.-E., Karvounis, M., Marinos, G., Theodorakopoulou, Z., Aloizou, E., Petsangourakis, G., Papakonstantinou, M., & Stoitsis, G. (2024). Comprehensive analysis of PFAS presence from environment to plate. npj Science of Food, 8, 80. https://doi.org/10.1038/s41538-024-00319-1
- Cordner, A., Goldenman, G., Birnbaum, L. S., Brown, P., Miller, M. F., Mueller, R., Patton, S., Salvatore, D. H., & Trasande, L. (2021). The true cost of PFAS and the benefits of acting now. Environmental Science & Technology, 55(15), 9630–9633. https://doi.org/10.1021/acs.est.1c03565
- Dehghani, M. H., Aghaei, M., Bashardoust, P., Rezvani Ghalhari, M., Nayeri, D., Malekpoor, M., Sheikhi, S., & Shi, Z. (2025). An insight into the environmental and human health impacts of per- and polyfluoroalkyl substances (PFAS): Exploring exposure pathways and their implications. Environmental Sciences Europe, 37, 81. https://doi.org/10.1186/s12302-025-01122-9
- DeLuca, N. M., Minucci, J. M., Mullikin, A., Slover, R., & Cohen Hubal, E. A. (2022). Human exposure pathways to poly- and perfluoroalkyl substances (PFAS) from indoor media: A systematic review. Environment International, 162, 107149. https://doi.org/10.1016/j.envint.2022.107149
- Kemper, J. A., Sharp, E., Yi, S., Leitao, E. M., Padhye, L. P., Kah, M., Chen, J. L.-Y., & Gobindlal, K. (2024). Public perceptions of per- and polyfluoroalkyl substances (PFAS): Psycho-demographic characteristics differentiating PFAS knowledge and concern. Journal of Cleaner Production, 442, 140866. https://doi.org/10.1016/j.jclepro.2024.140866
- Tachachartvanich, P., Singam, E. R. A., Durkin, K. A., Furlow, J. D., Smith, M. T., & La Merrill, M. A. (2022). In vitro characterization of the endocrine disrupting effects of per- and poly-fluoroalkyl substances (PFASs) on the human androgen receptor. Journal of Hazardous Materials, 429, 128243. https://doi.org/10.1016/j.jhazmat.2022.128243
- Bonato, M., Corrà, F., Bellio, M., Guidolin, L., Tallandini, L., Irato, P., & Santovito, G. (2020). PFAS environmental pollution and antioxidant responses: An overview of the impact on humans. International Journal of Environmental Research and Public Health, 17(21), 8020. https://doi.org/10.3390/ijerph17218020
- Kar, S., Sepúlveda, M. S., Roy, K., & Leszczynski, J. (2017). Endocrine-disrupting activity of per- and polyfluoroalkyl substances: Exploring combined approaches of ligand and structure-based modelling. Chemosphere, 184, 514–523. https://doi.org/10.1016/j.chemosphere.2017.06.024
- Coperchini, F., Teliti, M., Greco, A., Croce, L., & Rotondi, M. (2024). Per-polyfluoroalkyl substances (PFAS) as thyroid disruptors: Is there evidence for multi-transgenerational effects? Expert Review of Endocrinology & Metabolism, 19(4), 307–315. https://doi.org/10.1080/17446651.2024.2351885
- Birru, R. L., Liang, H.-W., Farooq, F., Bedi, M., Feghali, M., Haggerty, C. L., Mendez, D. D., Catov, J. M., Ng, C. A., & Adibi, J. J. (2021). A pathway level analysis of PFAS exposure and risk of gestational diabetes mellitus. Environmental Health, 20, 63. https://doi.org/10.1186/s12940-021-00740-z
- Voros, C., Athanasiou, D., Papapanagiotou, I., Mavrogianni, D., Varthaliti, A., Bananis, K., Athanasiou, A., Papadimas, G., & Gkirgkinoudis, A., et al. (2025). Molecular shadows of per- and polyfluoroalkyl substances (PFASs): Unveiling the impact of perfluoroalkyl substances on ovarian function, polycystic ovarian syndrome (PCOS), and in vitro fertilization (IVF) outcomes. International Journal of Molecular Sciences, 26(14), 6604. https://doi.org/10.3390/ijms26146604
- Xia, Z., Chen, S., Liu, Y., Li, J., Liu, X., Zhang, L., Xiang, Q., & Wu, Y. (2025). In utero exposure to per- and polyfluoroalkyl substances and neonatal sex hormone levels: Implications of endocrine disrupting effects during critical development windows. Environmental Chemistry and Ecotoxicology, 7, 1587–1597. https://doi.org/10.1016/j.enceco.2025.06.007
- Ismail, U. M., Elnakar, H., & Khan, M. F. (2023). Sources, fate, and detection of dust-associated perfluoroalkyl and polyfluoroalkyl substances (PFAS): A review. Toxics, 11(4), 335. https://doi.org/10.3390/toxics11040335
- Duru, C. I., Kang, D. H., & Sherchan, S. P. (2024). The trends of per- and polyfluoroalkyl substances (PFAS) in drinking water systems in Maryland, United States. Science of the Total Environment, 957, 177152. https://doi.org/10.1016/j.scitotenv.2024.177152
- Di Nisio, A., Lopez-Espinosa, M. J., & Foresta, C. (2022). Editorial: Emerging chemical risks for human health: Endocrine disruption by per- and polyfluorinated alkyl substances (PFAS). Frontiers in Endocrinology, 12, 813785. https://doi.org/10.3389/fendo.2021.813785
- Pascu, L. F., Petre, V. A., Cimpean, I. A., Paun, I., Pirvu, F., & Chiriac, F. L. (2025). Managing PFAS in sewage sludge: Exposure pathways, impacts, and treatment innovations. Journal of Xenobiotics, 15(4), 135. https://doi.org/10.3390/jox15040135
- Donley, N., Cox, C., Bennett, K., Temkin, A. M., Andrews, D. Q., & Naidenko, O. V. (2024). Forever pesticides: A growing source of PFAS contamination in the environment. Environmental Health Perspectives, 132(7), 075003. https://doi.org/10.1289/EHP13954
