Phthalates as Endocrine Disruptors: Health Risks of Phthalate Exposure
Table of Contents
- Abstract
- Introduction
- Reproductive Health: Male Effects
- Reproductive Health: Female Effects
- Developmental Impacts: The Vulnerable Foetus and Child
- Metabolic and Endocrine Disorders
- Neurodevelopmental Impacts
- Carcinogenic Potential
- Mechanisms of Multi-System Toxicity
- Vulnerable Populations and Critical Windows
- Special Exposure Scenarios
- Cumulative and Mixture Effects
- Conclusions and Implications
- References
Abstract
Phthalates are ubiquitous endocrine disrupting chemicals (EDCs) found throughout modern environments that pose significant and multifaceted threats to human health. As synthetic plasticizers that leach readily from consumer products, these compounds create unavoidable chronic exposure scenarios affecting populations across all life stages. Phthalates disrupt reproductive function in both males and females, interfere with foetal and child development, promote metabolic dysfunction including obesity and diabetes, alter thyroid hormone regulation, impair neurodevelopment, and carry potential carcinogenic properties. The anti-androgenic effects of phthalates manifest as reduced testosterone production, leading to developmental abnormalities in males including cryptorchidism, hypospadias, and the constellation of defects termed "phthalate syndrome." In females, exposure associates with endometriosis, disrupted ovarian function, and adverse pregnancy outcomes including preterm birth. Metabolic impacts include promotion of adipogenesis, insulin resistance, and metabolic syndrome. Neurodevelopmental consequences encompass cognitive deficits and motor skill impairments in children. Particularly concerning is the vulnerability of foetuses and neonates, who face exposure during critical developmental windows when hormonal disruption carries lifelong consequences. The mechanistic basis for these diverse effects lies in phthalates' ability to interfere with multiple hormonal signalling pathways, including steroid receptors, thyroid receptors, and metabolic regulators. These health risks underscore the urgent need for continued regulatory action, development of safer alternatives, and strategies to minimise exposure, particularly among vulnerable populations. This review examines the breadth of health risks associated with phthalate exposure, organised by physiological system and life stage.
Introduction
The recognition of phthalates as EDCs has evolved from initial concerns about isolated reproductive effects to an understanding of their capacity to disrupt human health across virtually every physiological system. These synthetic compounds, designed for industrial utility, have become inadvertent participants in the most fundamental processes of human biology, hormone signalling, development, metabolism, and cellular function [1; 2; 3].
What makes phthalates particularly insidious is not merely their toxicity at high doses, but their ability to exert adverse effects at environmentally relevant concentrations through disruption of hormonal homeostasis [4; 5; 6]. Unlike many environmental toxins that require substantial exposure to cause harm, phthalates can interfere with endocrine function at levels commonly encountered in daily life [7; 6; 8]. Furthermore, exposure begins before birth and continues throughout the lifespan, creating cumulative and potentially intergenerational consequences.
Understanding the risks posed by phthalate exposure requires appreciating both the specificity of certain phthalate effects, such as the anti-androgenic properties that particularly affect male development, and the systemic nature of endocrine disruption that creates cascading effects throughout the body.
Reproductive Health: Male Effects
Male Developmental Abnormalities and the Phthalate Syndrome
Male reproductive development represents one of the most extensively studied and clearly established areas of phthalate toxicity. The mechanistic basis centres on the anti-androgenic properties of several phthalates, particularly di(2-ethylhexyl) phthalate (DEHP) and dibutyl phthalate (DBP), which interfere with testosterone production during critical developmental windows [1; 9].
During foetal development, testosterone plays an essential role in masculinising the reproductive tract and external genitalia. Phthalate exposure during this sensitive period can produce a characteristic pattern of abnormalities termed "phthalate syndrome" in experimental models.
These developmental abnormalities share a common aetiology: disruption of foetal testosterone production by Leydig cells in the developing testes. Phthalates interfere with the enzymatic pathways necessary for testosterone synthesis, creating a state of relative androgen insufficiency during the precise windows when these hormones orchestrate male sexual differentiation [10]. Table 1 shows abnormalities and mechanistic basis in male development.
📊 Table 1: Male Developmental Abnormalities Associated with Phthalate Exposure
Abnormality | Description | Mechanistic basis | References |
Reduced anogenital distance (AGD) | Shorter distance between anus and genitals; biomarker of insufficient prenatal androgen exposure | Disruption of testosterone production during foetal development | 6; 9; 10 |
Cryptorchidism | Undescended testes; failure of normal testicular descent | Inadequate androgen signalling impairs descent; increased risk of infertility and cancer | 6; 9 |
Hypospadias | Urethral opening located along underside of penis rather than tip | Insufficient androgen action during critical developmental windows | 9; 10 |
Adult Male Fertility
The reproductive consequences of phthalate exposure extend beyond developmental abnormalities to affect fertility in adult men. Multiple studies have documented associations between phthalate exposure and various parameters of sperm quality [11].
The mechanisms underlying these effects likely involve both direct testicular toxicity and disruption of the hormonal signals that regulate spermatogenesis. Additionally, oxidative stress induced by phthalate exposure may contribute to cellular damage in developing sperm cells. Table 2 shows abnormalities and mechanistic basis associated with phthalate exposure in adult males.
📊 Table 2: Adult Male Reproductive Health Effects Associated with Phthalate Exposure
Abnormality | Mechanistic Basis | References |
Reduced sperm concentration and motility | Impaired spermatogenesis and sperm function; oxidative stress contributes to damage | 11 |
Abnormal sperm morphology | Structural defects in sperm linked to phthalate exposure | 11 |
DNA fragmentation | Genetic damage in sperm, increasing miscarriage risk and affecting offspring health | 11 |
Reproductive Health: Female Effects
Female reproductive health represents a critical domain of phthalate toxicity, with evidence showing that these chemicals disrupt ovarian function, fertility, and pregnancy outcomes. Conditions such as endometriosis and diminished ovarian reserve highlight how phthalates interfere with hormonal signalling and cellular processes essential for reproductive success [1; 8; 12]. The impacts extend beyond natural fertility to assisted reproduction, where higher phthalate exposure correlates with poorer IVF outcomes [13]. Together, these findings underscore the broad and clinically significant risks phthalates pose to women’s reproductive health. Table 3 shows abnormalities and mechanistic basis associated with phthalate exposure in female reproductive health.
📊 Table 3: Female Reproductive Health Effects of Phthalates
Outcome | Description | Mechanistic Basis | Key References |
Endometriosis | Growth of endometrial tissue outside uterus; causes pain, inflammation, fertility problems | Oestrogenic activity, inflammatory promotion, altered cell viability and proliferation | 1; 8; 12 |
Diminished ovarian reserve | Accelerated decline in viable eggs; earlier reproductive senescence | Disruption of folliculogenesis and ovarian maturation pathways | 13 |
Disrupted hormone production | Altered oestrogen and progesterone synthesis; irregular menstrual cycles and impaired pregnancy maintenance | Interference with steroidogenic pathways in ovarian cells | 13 |
Impaired IVF outcomes | Poorer assisted reproduction outcomes: fewer mature eggs, lower fertilisation, reduced implantation success | Phthalate-induced disruption of ovarian function and hormone signalling | 13 |
Pregnancy Outcomes
Pregnancy outcomes represent one of the most critical domains in which phthalate exposure exerts adverse effects, given the dual impact on maternal health and foetal development. These synthetic chemicals readily cross the placental barrier, exposing the developing foetus during sensitive windows when hormonal disruption can have lifelong consequences. Epidemiological studies increasingly link maternal phthalate exposure to complications such as preterm birth, pregnancy loss, and low birth weight, underscoring their role as significant environmental risk factors [13; 14]. Understanding these associations is essential, as they highlight both the immediate risks to neonatal survival and the long-term implications for child health and disease susceptibility.
The mechanisms linking phthalates to preterm birth likely involve inflammatory pathways and disruption of the hormonal signals that maintain pregnancy [13; 14]. Phthalates may trigger premature activation of labour-inducing pathways or interfere with progesterone signalling that normally suppresses uterine contractions. Table 4 shows pregnancy outcomes linked to phthalate exposure.
📊 Table 4: Pregnancy Outcomes Linked to Phthalate Exposure
Outcome | Description | Mechanistic Basis | Key References |
Preterm birth | Delivery before 37 weeks; major cause of infant morbidity and mortality | Inflammatory pathways; disruption of progesterone signalling; premature activation of labour-inducing pathways; specific metabolites showing particularly strong associations include MEP (monoethyl phthalate), MECPP (mono-(2-ethyl-5-carboxypentyl) phthalate), MBzP, and DEHP metabolites. | 13; 14 |
Pregnancy loss | Miscarriage and stillbirth; associations vary by timing and exposure level | Hormonal disruption; inflammatory stress; impaired maintenance of pregnancy | 9; 13 |
Low birth weight | Infants born small for gestational age, even at term; linked to long-term metabolic and cardiovascular risks | Impaired placental function; disrupted nutrient and hormone signalling | 9; 13 |
Developmental Impacts: The Vulnerable Foetus and Child
Developmental impacts represent one of the most critical areas of concern in phthalate toxicity, as exposure during foetal and early childhood stages can have lifelong consequences. The foetus is uniquely vulnerable because hormonal signals must be precisely timed to orchestrate organ and tissue development, and phthalates readily cross the placental barrier to disrupt these processes [2; 9; 15]. Neonates in intensive care face an unfortunate paradox: the medical devices essential for their survival often contain phthalates, exposing them during a period of heightened susceptibility [16]. Beyond infancy, children continue to encounter elevated exposure through everyday behaviours and environments, making early life a continuum of vulnerability to phthalate-related health risks [9]. Table 5 shows the impact of phthalate exposure in foetus, neonates, and children.
📊 Table 5: Developmental Impacts of Phthalate Exposure in Foetus, Neonates, and Children
Life Stage | Exposure Source | Outcome / Risk | Mechanistic Basis | Key References |
In utero (foetus) | Maternal exposure; placental transfer; confirmed in amniotic fluid | Lifelong programming of health trajectories; structural and functional alterations | Endocrine disruption of testosterone, thyroid, and other hormonal pathways; altered tissue development | 2; 9; 15 |
Neonatal intensive care | Medical devices (IV lines, feeding tubes, respiratory equipment, transfusion bags) | High phthalate doses during critical developmental windows; long-term health compromise | DEHP leaching from PVC plastics; immature hepatic metabolism and blood-brain barrier increase vulnerability | 16 |
Childhood | Toys, household dust, food intake relative to body weight | Neurodevelopmental, reproductive, and metabolic risks during ongoing organ maturation | Hand-to-mouth behaviours; environmental accumulation; endocrine disruption during growth | 3 3; 9 |
Metabolic and Endocrine Disorders
Phthalates exert profound effects on metabolic and endocrine systems, acting as “obesogens” that disrupt weight regulation, glucose metabolism, and thyroid hormone function [17]. Their influence extends beyond simple caloric balance, interfering with hormonal signalling pathways and cellular processes that control adipogenesis, insulin sensitivity, and thyroid regulation. The following tables (6 - 10) summarise the major metabolic and endocrine disorders associated with phthalate exposure, their mechanisms, and supporting evidence.
📊 Table 7: Phthalates as Obesogens
Effect | Description / Clinical Impact | Mechanistic Basis | Key References |
Adipogenesis promotion | Increased differentiation of precursor cells into mature adipocytes | Activation of PPARγ, master regulator of adipocyte differentiation | 18 |
Altered lipid metabolism | Disrupted synthesis, storage, and breakdown of lipids | Dysregulation of metabolic pathways → fat accumulation | 6 |
Endocrine disruption in weight regulation | Dysregulated appetite, energy expenditure, and metabolic rate | Interference with thyroid hormones, sex steroids, and other endocrine signals | 13 |
Epidemiological evidence | Higher BMI, waist circumference, obesity across age groups; prenatal exposure linked to childhood adiposity | Developmental programming of metabolic dysfunction | 6; 18 |
📊 Table 8: Metabolic Syndrome
Effect | Description / Clinical Impact | Mechanistic Basis | Key References |
Abdominal obesity | Central fat accumulation, cardiovascular risk | Endocrine disruption and altered lipid metabolism | 18 |
Elevated blood pressure | Hypertension as part of metabolic syndrome cluster | Vascular effects of endocrine disruption and inflammation | 6 |
Dyslipidemia | Abnormal cholesterol and triglyceride levels | Altered lipid metabolism and PPAR activation | 17 |
Insulin resistance | Reduced cellular responsiveness to insulin | Obesity, ectopic fat accumulation, oxidative stress | 6 |
📊 Table 9: Diabetes and Insulin Resistance
Effect | Description / Clinical Impact | Mechanistic Basis | Key References |
Insulin resistance | Impaired insulin signaling; reduced glucose uptake | Disruption of insulin receptor cascade; oxidative stress; ectopic fat accumulation | 2; 19 |
Beta cell dysfunction | Damage to pancreatic beta cells; impaired insulin production | Oxidative stress; apoptosis of beta cells | 19 |
PPAR activation | Dysregulated glucose metabolism | Chronic activation of PPAR pathways affecting insulin sensitivity | 2 |
Epidemiological evidence | Higher diabetes risk, elevated fasting glucose, HbA1c, insulin resistance | Early life exposure programs long-term metabolic dysfunction | 2 |
📊 Table 10: Thyroid Dysfunction
Effect | Description / Clinical Impact | Mechanistic Basis | Key References |
Decreased serum thyroxine (T₄) | Lower circulating thyroid hormone levels | Disruption of synthesis, transport, uptake, and metabolism | 20 |
Interference with synthesis | Impaired enzymatic pathways for thyroid hormone production | Direct disruption of thyroid gland function | 20 |
Altered hormone transport | Reduced binding to carrier proteins; impaired delivery to tissues | Competition with binding proteins | 20 |
Disrupted uptake/metabolism | Impaired cellular utilization of thyroid hormones | Endocrine disruption at cellular level | 20 |
Axis disruption | Altered hypothalamic-pituitary-thyroid feedback regulation | Phthalate interference with hormone receptors and signaling pathways | 20 |
Metabolic Continuum
Phthalate exposure creates a metabolic continuum, beginning with obesogenic effects and progressing through metabolic syndrome, diabetes, and thyroid dysfunction. These interconnected outcomes illustrate how endocrine disruption amplifies risk across multiple systems, programming long-term health trajectories from early life onward (Table 11).
📊 Table 11: Metabolic Continuum of Phthalate Exposure
Stage / Disorder | Clinical Impact | Mechanistic Basis | Key References |
Obesity (Obesogen effect) | Increased BMI, waist circumference, adiposity in children and adults | Activation of PPARγ → adipogenesis; altered lipid metabolism; endocrine disruption of appetite and energy balance | 6 ; 13; 18 |
Metabolic Syndrome | Cluster of risk factors: abdominal obesity, hypertension, dyslipidemia, insulin resistance | Dose-dependent metabolic dysfunction; endocrine disruption; chronic inflammation | 6 ; 17; 18 |
Type 2 Diabetes and Insulin Resistance | Elevated fasting glucose, HbA1c, impaired insulin signaling, beta cell dysfunction | Disruption of insulin receptor cascade; oxidative stress; ectopic fat accumulation; apoptosis of beta cells | 2; 19 |
Thyroid Dysfunction | Decreased serum thyroxine (T₄); impaired metabolism, growth, brain development | Disrupted synthesis, transport, uptake, and feedback regulation of thyroid hormones | 20 |
Neurodevelopmental Impacts
Neurodevelopmental impacts represent one of the most concerning consequences of phthalate exposure, as the developing brain is highly sensitive to endocrine disruption and neurotoxic insults. Hormonal signals such as thyroid hormones [9] and sex steroids [21; 22] are essential for neuronal growth, migration, synapse formation, and myelination. When disrupted during critical developmental windows, these processes can be permanently altered, leading to cognitive, motor, and behavioural deficits. Emerging evidence links prenatal and early childhood phthalate exposure to measurable impairments in learning, motor coordination, and behaviour, underscoring the lifelong implications of early exposure [9] (Table 12). The underlying mechanisms, ranging from thyroid hormone disruption to oxidative stress and neurotransmitter interference, are summarised in Table 13: Mechanisms of Neurotoxicity.
📊 Table 12: Neurodevelopmental Impacts of Phthalate Exposure
Outcome | Description / Clinical Impact | Mechanistic Basis | Key References |
Cognitive deficits | Poorer performance on intelligence, memory, and learning assessments | Thyroid hormone disruption; altered neurotransmitter systems | 9; 23 |
Visual reception impairments | Problems with visual processing and visual-motor integration, especially linked to MEP exposure | Endocrine disruption affecting sensory integration pathways | 9 |
Fine motor skill deficits | Impaired hand coordination and precision tasks; associated with DEHTP metabolites | Direct neuronal effects; oxidative stress damaging motor control circuits | 23 |
Behavioural problems | Increased attention difficulties and externalizing behaviours | Altered dopaminergic and neurotransmitter systems; oxidative stress | 23 |
📊 Table 13: Mechanisms of Neurotoxicity
Mechanism | Description | Implications |
Thyroid hormone disruption | Impaired thyroid function during critical developmental periods | Deficits in brain maturation, cognition, and motor skills |
Direct neuronal effects | Toxicity to neurons and glial cells; impaired proliferation and differentiation | Structural and functional brain abnormalities |
Oxidative stress | Damage to lipids, proteins, and DNA in developing brain cells | Long-term impairment of neuronal survival and signalling |
Altered neurotransmitter systems | Disruption of dopaminergic and other pathways critical for cognition and behaviour | Behavioural problems, attention deficits, learning difficulties |
Carcinogenic Potential
The carcinogenic potential of phthalates has become an increasing focus of regulatory and scientific scrutiny. While animal studies demonstrate elevated tumour incidence with exposure, human evidence remains more limited but biologically plausible. The Environmental Protection Agency classifies DEHP as a probable human carcinogen (Group B2), while the International Agency for Research on Cancer (IARC) classifies it as possibly carcinogenic to humans (Group 2B) [24]. Epidemiological findings highlight associations with breast cancer, thyroid cancer, and broader risks linked to air pollution, underscoring the need to understand the mechanisms, ranging from oestrogenic activity to oxidative stress, that may drive carcinogenesis.
📊 Table 14: Carcinogenic Potential of Phthalates
Cancer Type / Risk | Description / Evidence | Mechanistic Basis | Key References |
Classification (DEHP) | EPA: Probable human carcinogen (Group B2); IARC: Possibly carcinogenic (Group 2B) | Evidence from animal studies showing increased tumor incidence; limited human data | 24 |
Breast Cancer | Consistent epidemiological associations with DBP and DEHP exposure; increased risk of invasive breast cancer | Estrogenic activity, chronic inflammation, epigenetic alterations, oxidative stress and DNA damage | 25; 26 |
Thyroid Cancer | Limited direct human evidence; strong biological plausibility due to thyroid disruption | Endocrine disruption of thyroid hormone synthesis and tissue function | 24 |
Air Pollution–related risk | DEHP contributes to endocrine-disrupting potential of PM₂.₅; may elevate lung cancer risk | Particulate phthalates in ambient air; oxidative stress and endocrine disruption | 27 |
Mechanisms of Multi-System Toxicity
Phthalates exert toxic effects across multiple organ systems, with endocrine disruption serving as the central mechanism that links diverse health outcomes. Hormones act as master regulators of physiology, and interference with their signalling cascades produces widespread consequences for reproduction, metabolism, neurodevelopment, and carcinogenesis [13; 21]. In addition to disrupting hormone pathways, phthalates induce oxidative stress and chronic inflammation, compounding their impact by damaging cellular structures and promoting disease processes. Together, these mechanisms explain how phthalates function as multi‑system toxicants with far‑reaching implications for human health (Table 15).
📊 Table 15: Mechanisms of Multi-System Toxicity of Phthalates
Mechanism | Description / Action | Systemic Consequences | Key References |
Receptor agonism/antagonism | Binding to hormone receptors (ER, AR, nuclear receptors), activating or blocking signals | Disrupted reproductive development, altered sexual differentiation, hormone-dependent cancers | 5; 6 |
Altered hormone synthesis | Interference with steroidogenic enzymes; reduced testosterone and oestrogen production | Anti-androgenic effects, reproductive abnormalities, impaired fertility | 4; 6 |
Modified hormone metabolism | Changes in hormone breakdown and inactivation | Prolonged or diminished hormonal signaling; dysregulated endocrine homeostasis | 5 |
Disrupted hormone transport | Competition for binding proteins or altered protein expression | Impaired hormone delivery to target tissues; systemic endocrine imbalance | 4 |
PPAR activation | Activation of PPARγ affecting lipid and glucose metabolism | Adipogenesis, obesity, insulin resistance, metabolic syndrome | 6 |
Oxidative stress | Excess reactive oxygen species damaging lipids, proteins, DNA | Cellular injury, mutagenesis, neurotoxicity, carcinogenesis | 2 |
Inflammatory pathways | Increased production of pro-inflammatory cytokines | Chronic inflammation linking endometriosis, metabolic syndrome, cancer | 2 |
Vulnerable Populations and Critical Windows
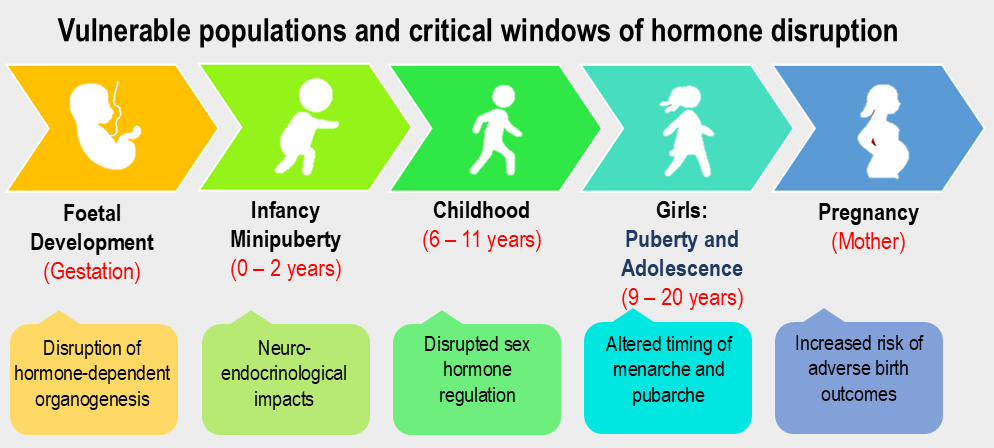
Phthalate exposure exerts disproportionate effects during critical developmental windows, when hormonal and physiological systems are undergoing rapid change. From foetal development through infancy, childhood, puberty, adolescence, and pregnancy, these stages represent periods of heightened vulnerability [9]. Endocrine disruption during these windows can permanently alter reproductive, metabolic, and neurodevelopmental trajectories. Table 16 integrates evidence on vulnerable populations with detailed impacts on hormonal and developmental timing.
📊Table 16: Timeline of Vulnerable Populations and Critical Windows of Phthalate Sensitivity
Life Stage | Age Range | Key Vulnerabilities | Dominant Impacts | Key References |
Foetal Development | Gestation | Hormone-dependent organogenesis; metabolic and neurodevelopmental programming | Permanent alterations in reproductive anatomy, brain development, and metabolic regulation | 2; 9; 15 |
Infancy / Minipuberty (boys) | 0–2 years (0.5–7 mo) | Neuroendocrine activation of HPG axis; high exposure via hand-to-mouth behaviour | Altered LH, AMH, INSL3; lower testosterone/LH ratio; endocrine programming effects | 9; 28 |
Childhood | 6–11 years | Ongoing hormonal regulation; early pubertal transitions | Disrupted sex hormone levels (E2, TT, FAI); elevated SHBG | 29 |
Puberty & Adolescence (girls) | 9–20 years | Hormonal surges; reproductive maturation; maternal exposure effects | Precocious puberty, altered menarche/pubarche timing; reduced AMH; lower testosterone | 6; 13; 30 |
Puberty & Adolescence (boys) | 12–20 years | Hormonal surges; reproductive maturation | Lower E2, TT, FAI; inverse associations with BPA, MBzP, MiBP | 29 |
Pregnancy (mother) | Gestation | Altered maternal hormone levels and metabolic adjustments | Increased risk of preterm birth, low birth weight, pregnancy loss | — |
Timeline of Vulnerable Populations and Critical Windows
The timeline in Figure 1 highlights vulnerable populations and critical windows of phthalate sensitivity, spanning from fetal development through infancy, childhood, puberty, adolescence, and pregnancy. Each stage represents a period of heightened hormonal and physiological vulnerability, where endocrine disruption can permanently alter reproductive, metabolic, and neurodevelopmental outcomes.
Special Exposure Scenarios
While phthalate exposure is widespread in the general population, certain scenarios result in acute or disproportionately high exposure, often during periods of heightened physiological vulnerability. These special exposure contexts, such as neonatal intensive care, medical procedures, and occupational environments, can deliver phthalates at doses far exceeding typical environmental levels. The combination of high exposure intensity and biological sensitivity underscores the need for targeted risk mitigation in these settings. Table 17 summarises key high-risk scenarios and their implications.
📊 Table 17: Special Exposure Scenarios for Phthalates
Exposure Scenario | Population Affected | Exposure Characteristics | Health Implications | Key References |
Neonatal Intensive Care (NICU) | Premature and critically ill infants | High-dose exposure via plastic medical devices (tubing, catheters, IV bags) during critical developmental window | Endocrine disruption, neurodevelopmental compromise, long-term metabolic and reproductive effects | 16 |
Medical Procedures | Patients undergoing transfusions, dialysis, or plastic-intensive interventions | Acute phthalate exposure from blood bags, tubing, and dialysis membranes | Hormonal imbalance, oxidative stress, potential reproductive and metabolic consequences |
|
Occupational Exposure | Workers in plastics, cosmetics, and chemical manufacturing sectors | Chronic high-level exposure via inhalation, dermal contact, and environmental contamination | Increased risk of reproductive dysfunction, hormone-related cancers, and systemic toxicity |
|
Cumulative and Mixture Effects
The Cocktail Problem
Humans are never exposed to phthalates in isolation. Rather, exposure involves mixtures of multiple phthalates simultaneously, along with other endocrine-disrupting chemicals such as bisphenols, pesticides, and persistent organic pollutants. This "cocktail effect" complicates risk assessment, as chemicals may interact in additive, synergistic, or antagonistic ways.
Some phthalates may act through similar mechanisms, potentially producing additive effects, the combined impact equals the sum of individual effects. However, synergistic interactions are also possible, where the combined effect exceeds the sum of individual contributions. Understanding these mixture effects remains a major challenge in environmental health research.
Cumulative Exposure Across the Lifespan
The chronic nature of phthalate exposure creates cumulative risks. While individual phthalates have short biological half-lives and are rapidly metabolised, continuous exposure means the body never fully clears these compounds. This creates a scenario where tissues and organs face constant endocrine disruption rather than isolated insults.
Moreover, effects may accumulate across the lifespan. Prenatal exposure may programme metabolic dysfunction that manifests in childhood. Childhood exposure may impair reproductive development in ways that affect adult fertility. Adult exposure may accelerate age-related diseases. The full impact of lifelong phthalate exposure may thus exceed what would be predicted from studying any single life stage in isolation.
Conclusions and Implications
The breadth and severity of health risks associated with phthalate exposure present a sobering picture of how industrial chemicals designed for practical utility can become widespread threats to human health. From reproductive dysfunction and developmental abnormalities to metabolic disease, neurodevelopmental deficits, and potential carcinogenicity, phthalates demonstrate capacity to disrupt virtually every physiological system.
Several themes emerge from this comprehensive review:
- Multi-system impacts: Phthalates do not target a single organ or process but rather disrupt fundamental hormonal signalling pathways that coordinate physiology across the body. This creates cascading effects where dysfunction in one system amplifies problems in others.
- Developmental vulnerability: The most concerning risks involve exposure during foetal development, infancy, and childhood; periods when endocrine disruption can permanently alter developmental trajectories. The concept that prenatal and early-life exposures program lifelong health outcomes underscores the urgency of protecting vulnerable populations.
- Low-dose effects: Unlike traditional toxins requiring substantial exposure to cause harm, phthalates exert adverse effects at environmentally relevant concentrations through endocrine disruption. This challenges traditional dose-response assumptions and suggests that even seemingly low exposures may be harmful.
- Ubiquitous and unavoidable exposure: The widespread presence of phthalates in consumer products and the environment creates scenarios where avoiding exposure entirely is impossible. This shifts the challenge from individual behaviour change to systemic solutions involving product reformulation, regulatory action, and development of safer alternatives.
- Gaps in knowledge: Despite extensive research, significant uncertainties remain regarding long-term health consequences, mixture effects, transgenerational impacts, and the comparative safety of alternative plasticizers. Continued research is essential to fully characterise risks and guide regulatory decisions.
The health risks documented in this review underscore the urgent need for action on multiple fronts: continued regulatory restrictions on high-concern phthalates, development and validation of safer alternatives, improved exposure assessment to identify high-risk populations and scenarios, and strategies to minimise exposure, particularly among pregnant women, infants, and children. Only through comprehensive approaches addressing both individual exposure reduction and systemic changes in how phthalates are regulated and used can we hope to mitigate the significant health risks these ubiquitous chemicals pose to current and future generations.
References
- Interdonato, L., Siracusa, R., Fusco, R., Cuzzocrea, S., & Di Paola, R. (2023). Endocrine disruptor compounds in environment: Focus on women’s reproductive health and endometriosis. International Journal of Molecular Sciences, 24(6), 5682. https://doi.org/10.3390/ijms24065682
- Mariana, M., & Cairrao, E. (2023). The relationship between phthalates and diabetes: A review. Metabolites, 13(6), https://doi.org/10.3390/metabo13060746
- Wang, Y., & Qian, H. (2021). Phthalates and their impacts on human health. Healthcare, 9(5), 603. https://doi.org/10.3390/healthcare9050603
- Li, Y., Yang, H., He, W., & Li, Y. (2023). Human endocrine-disrupting effects of phthalate esters through adverse outcome pathways: A comprehensive mechanism analysis. International Journal of Molecular Sciences, 24(17), 13548. https://doi.org/10.3390/ijms241713548
- Martínez-Pinna, J., Sempere-Navarro, R., Medina-Gali, R. M., Fuentes, E., Quesada, I., Sargis, R. M., Trasande, L., & Nadal, A. (2023). Endocrine disruptors in plastics alter β-cell physiology and increase the risk of diabetes mellitus. American Journal of Physiology-Endocrinology and Metabolism, 324(4), E488–E505. https://doi.org/10.1152/ajpendo.00068.2023
- Predieri, B., Iughetti, L., Bernasconi, S., & Street, M. E. (2022). Endocrine disrupting chemicals’ effects in children: What we know and what we need to learn. International Journal of Molecular Sciences, 23(19), 11899. https://doi.org/10.3390/ijms231911899
- Martin, L., Zhang, Y., First, O., Mustieles, V., Dodson, R., Rosa, G., Coburn-Sanderson, A., Adams, C. D., & Messerlian, C. (2022). Lifestyle interventions to reduce endocrine-disrupting phthalate and phenol exposures among reproductive age men and women: A review and future steps. Environment International, 170, 107576. https://doi.org/10.1016/j.envint.2022.107576
- Ribeiro, B., Mariana, M., Lorigo, M., Oliani, D., Ramalhinho, A. C., & Cairrao, E. (2024). Association between the exposure to phthalates and the risk of endometriosis: An updated review. Biomedicines, 12(8), 1932. https://doi.org/10.3390/biomedicines12081932
- Lucaccioni, L., Trevisani, V., Passini, E., Righi, B., Plessi, C., Predieri, B., & Iughetti, L. (2021). Perinatal exposure to phthalates: From endocrine to neurodevelopment effects. International Journal of Molecular Sciences, 22(8), 4063. https://doi.org/10.3390/ijms22084063
- Sharpe, R. M. (2024). Endocrine disruption and male reproductive disorders: Unanswered questions. Human Reproduction, 39(9), 1879–1888. https://doi.org/10.1093/humrep/deae143
- Virant-Klun, I., Imamovic-Kumalic, S., & Pinter, B. (2022). From oxidative stress to male infertility: Review of the associations of endocrine-disrupting chemicals (bisphenols, phthalates, and parabens) with human semen quality. Antioxidants, 11, https://doi.org/10.3390/antiox11081617
- Chitakwa, N., Alqudaimi, M., Sultan, M., & Wu, D. (2024). Plastic-related endocrine disrupting chemicals significantly related to the increased risk of estrogen-dependent diseases in women. Environmental Research, 252, 118966. https://doi.org/10.1016/j.envres.2024.118966
- Basso, C. G., de Araújo-Ramos, A. T., & Martino-Andrade, A. J. (2022). Exposure to phthalates and female reproductive health: A literature review. Reproductive Toxicology, 109, 61–79. https://doi.org/10.1016/j.reprotox.2022.02.006
- Wu, Y., Wang, J., Wei, Y., Chen, J., Kang, L., Long, C., Wu, S., Shen, L., & Wei, G. (2022). Maternal exposure to endocrine disrupting chemicals (EDCs) and preterm birth: A systematic review, meta-analysis, and meta-regression analysis. Environmental Pollution, 292, https://doi.org/10.1016/j.envpol.2021.118264
- Bräuner, E. V., Uldbjerg, C. S., Lim, Y.-H., Gregersen, L. S., Krause, M., Frederiksen, H., & Andersson, A.-M. (2022). Presence of parabens, phenols and phthalates in paired maternal serum, urine and amniotic fluid. Environment International, 158, 106987. https://doi.org/10.1016/j.envint.2021.106987
- Panneel, L., Cleys, P., Poma, G., Ait Bamai, Y., Jore. ns, P. G., Covaci, A., & Mulder, A. (2024). Ongoing exposure to endocrine disrupting phthalates and alternative plasticizers in neonatal intensive care unit patients. Environment International, 186, https://doi.org/10.1016/j.envint.2024.108605
- Biemann, R., Blüher, M., & Isermann, B. (2021). Exposure to endocrine-disrupting compounds such as phthalates and bisphenol A is associated with an increased risk for obesity. Best Practice & Research Clinical Endocrinology & Metabolism, 35, https://doi.org/10.1016/j.beem.2021.101546
- Dalamaga, M., Kounatidis, D., Tsilingiris, D., Vallianou, N. G., Karampela, I., Psallida, S., & Papavassiliou, A. G. (2024). The role of endocrine disruptors bisphenols and phthalates in obesity: Current evidence, perspectives and controversies. International Journal of Molecular Sciences, 25, https://doi.org/10.3390/ijms25010675
- Hinault, C., Caroli-Bosc, P., Bost, F., & Chevalier, N. (2023). Critical overview on endocrine disruptors in diabetes mellitus. International Journal of Molecular Sciences, 24(5), 4537. https://doi.org/10.3390/ijms24054537
- Gao, Q., Song, Y., Jia, Z., Huan, C., Cao, Q., Wang, C., Mao, Z., & Huo, W. (2024). Association of exposure to a mixture of phenols, parabens, and phthalates with altered serum thyroid hormone levels and the roles of iodine status and thyroid autoantibody status: A study among American adults. Ecotoxicology and Environmental Safety, 282, 116754. https://doi.org/10.1016/j.ecoenv.2024.116754
- Stathori, G., Hatziagapiou, K., Mastorakos, G., Vlahos, N. F., Charmandari, E., & Valsamakis, G. (2024). Endocrine-disrupting chemicals, hypothalamic inflammation and reproductive outcomes: A review of the literature. International Journal of Molecular Sciences, 25(21), 11344. https://doi.org/10.3390/ijms252111344
- Zhang, Y., Yang, Y., Tao, Y., Guo, X., Cui, Y., & Li, Z. (2023). Phthalates (PAEs) and reproductive toxicity: Hypothalamic-pituitary-gonadal (HPG) axis aspects. Journal of Hazardous Materials, 459, https://doi.org/10.1016/j.jhazmat.2023.132182
- Sotelo-Orozco, J., Calafat, A. M., Botelho, J. C., Schmidt, R. J., Hertz-Picciotto, I., & Bennett, D. H. (2024). Exposure to endocrine disrupting chemicals including phthalates, phenols, and parabens in infancy: Associations with neurodevelopmental outcomes in the MARBLES study. International Journal of Hygiene and Environmental Health, 261, https://doi.org/10.1016/j.ijheh.2024.114425
- Alsen, M., Sinclair, C., Cooke, P., Ziadkhanpour, K., Genden, E., & van Gerwen, M. (2021). Endocrine disrupting chemicals and thyroid cancer: An overview. Toxics, 9(1), 14. https://doi.org/10.3390/toxics9010014
- Benoit, L., Tomkiewicz, C., Bortoli, S., Bats, A. S., Coumoul, X., & Koual, M. (2025). Role of phthalates in breast cancer initiation, progression and drug resistance: A scoping review and recommendations. Toxicology Letters, 413, 111721. https://doi.org/10.1016/j.toxlet.2025.111721
- Mukherjee Das, A., Gogia, A., Garg, M., Elaiyaraja, A., Arambam, P., Mathur, S., Babu-Rajendran, R., Deo, S. V. S., Kumar, L., Das, B. C., & Janardhanan, R. (2022). Urinary concentration of endocrine-disrupting phthalates and breast cancer risk in Indian women: A case-control study with a focus on mutations in phthalate-responsive genes. Cancer Epidemiology, 79, 102188. https://doi.org/10.1016/j.canep.2022.102188
- Zhou, Q., Chen, J., Zhang, J., Zhou, F., Zhao, J., Wei, X., Zheng, K., Wu, J., Li, B., & Pan, B. (2022). Toxicity and endocrine-disrupting potential of PM2.5: Association with particulate polycyclic aromatic hydrocarbons, phthalate esters, and heavy metals. Environmental Pollution, 292, https://doi.org/10.1016/j.envpol.2021.118349
- Müller, M. L., Busch, A. S., Ljubicic, M. L., Upners, E. N., Fischer, M. B., Hagen, C. P., Albrethsen, J., Frederiksen, H., Juul, A., & Andersson, A.-M. (2023). Urinary concentration of phthalates and bisphenol A during minipuberty is associated with reproductive hormone concentrations in infant boys. International Journal of Hygiene and Environmental Health, 250, https://doi.org/10.1016/j.ijheh.2023.114166
- Hu, P., Pan, C., Su, W., Vinturache, A., Hue, Y., Dong, X., & Ding, G. (2022). Associations between exposure to a mixture of phenols, parabens, and phthalates and sex steroid hormones in children 6–19 years from NHANES, 2013–2016. Science of the Total Environment, 822, https://doi.org/10.1016/j.scitotenv.2022.153548
- Arrigo, F., Impellitteri, F., Piccione, G., & Faggio, C. (2023). Phthalates and their effects on human health: Focus on erythrocytes and the reproductive system. Comparative Biochemistry and Physiology Part C: Toxicology & Pharmacology, 270, https://doi.org/10.1016/j.cbpc.2023.109645
