Phthalates: Introducing Ubiquitous Human Health Disruptors
Table of Contents
- Abstract
- Introduction
- A Brief History of Phthalates and Regulatory Response
- Applications and Sources of Exposure
- Classification by Molecular Weight
- Routes of Human Exposure
- Vulnerable Populations
- Metabolism and Biological Fate
- Mechanisms of Endocrine Disruption
- Health Impacts Across the Lifespan
- A Systems Perspective on Phthalate Toxicity
- Conclusion
- References
Abstract
Phthalates, synthetic esters of phthalic acid, represent one of the most widespread classes of endocrine disrupting chemicals in modern environments. First introduced as industrial plasticizers in the 1920s, these compounds have become ubiquitous in consumer products ranging from food packaging and medical devices to cosmetics and household materials. Their non-covalent bonding to host materials facilitates ready leaching into the environment, creating scenarios of chronic, unavoidable human exposure that begin in utero and persist throughout life. Phthalates disrupt hormonal homeostasis by interfering with steroid hormone receptors, thyroid signalling, and metabolic regulation pathways. Health impacts span reproductive dysfunction in both sexes, increased risks of preterm birth, metabolic disorders including obesity and type 2 diabetes, thyroid disruption, neurodevelopmental deficits in children, and potential carcinogenicity. Vulnerable populations, particularly foetuses, neonates in intensive care, and young children, face disproportionate exposure and risk. Despite evolving regulations in North America and Europe that have restricted certain high-concern phthalates, exposure remains widespread, and replacement plasticizers raise new questions about comparative safety.
This review synthesizes current understanding of phthalate history, sources, exposure pathways, metabolic fate, and multi-system health impacts, establishing a foundation for understanding these chemicals as paradigmatic endocrine disruptors with far-reaching implications for human health across the lifespan.
Introduction
Phthalates represent one of the most pervasive chemical exposures in modern life. As esters of phthalic acid, these synthetic compounds have become integral to contemporary manufacturing, embedded in plastics, personal care products, food packaging, and countless consumer goods. Their versatility and low cost have ensured their widespread adoption, yet this very ubiquity comes at a concerning cost to human health [1; 2]. Recognised globally as endocrine-disrupting chemicals (EDCs), phthalates interfere fundamentally with hormonal signalling pathways that regulate reproduction, metabolism, development, and numerous other physiological processes [3; 2; 4; 5].
The insidious nature of phthalate exposure lies not only in its breadth but also in its persistence. These compounds leach readily into the environment, contaminating air, water, soil, and food chains, thereby creating a scenario of chronic, largely unavoidable human exposure. This exposure begins in utero, with measurable levels detected in maternal and cord blood, and continues throughout the lifespan, influencing health trajectories from infancy to adulthood [4; 6; 7]. The combination of widespread use, environmental mobility, and biological impact underscores why phthalates have become emblematic of the broader challenge posed by endocrine disruptors in modern society.
A Brief History of Phthalates and Regulatory Response
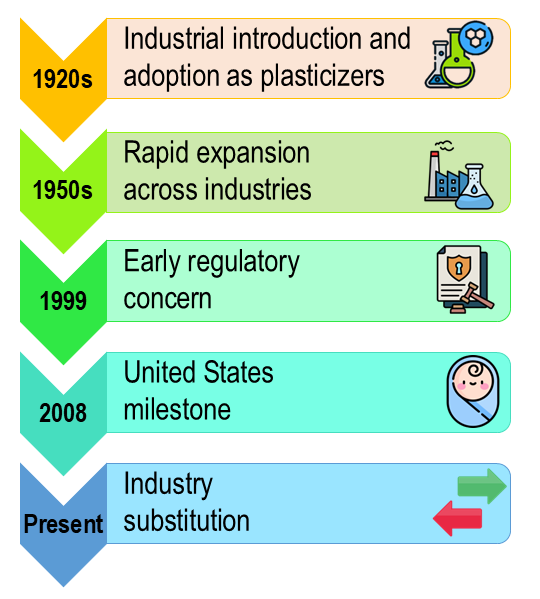
The story of phthalates begins in the early 20th century, when industrial chemists first synthesized these compounds as esters of phthalic acid. By the 1920s, phthalates were introduced into manufacturing processes, and by the 1930s they had found their niche application as plasticizers, chemicals added to plastics to impart flexibility, durability, and resilience. Their compatibility with polyvinyl chloride (PVC) proved especially transformative, enabling the production of a wide range of consumer and industrial goods, from flooring and cables to medical tubing and packaging materials [2; 8; 9]. This functional utility led to rapid adoption across industries, and by the latter half of the century, phthalates had become virtually omnipresent in manufactured goods, symbolising the chemical revolution that reshaped modern consumer culture.
As production and use expanded, so too did scientific concern about potential health consequences. Early toxicological studies hinted at reproductive and developmental effects, but it was not until the 1990s that regulatory bodies began to act decisively. The regulatory response commenced in earnest in 1994, when Health Canada classified di(2-ethylhexyl) phthalate (DEHP) as toxic to human health, citing evidence of carcinogenicity and endocrine disruption [10]. This designation marked a turning point in how authorities viewed these chemicals, shifting them from benign industrial additives to substances warranting precautionary oversight.
The European Union (EU) followed suit with restrictions in 1999, mandating that six plasticizers, including DEHP and dibutyl phthalate (DBP), not exceed 0.1% content in certain products, particularly those with direct consumer contact [1]. This regulatory framework reflected growing recognition of phthalates as endocrine-disrupting chemicals (EDCs) and set the stage for more comprehensive bans. In the United States, the 2008 Consumer Product Safety Improvement Act represented a milestone in protecting vulnerable populations, banning DEHP, DBP, and benzyl butyl phthalate (BBP) in children’s toys and childcare articles at concentrations above 0.1% [5]. These measures underscored the heightened concern for developmental exposure, given evidence that phthalates could interfere with hormonal signalling during critical windows of growth.
Subsequent EU actions further strengthened restrictions, classifying BBP, diisobutyl phthalate (DiBP), DBP, and DEHP as substances of very high concern under the REACH framework. Their presence in plastics was limited to less than 0.1% by weight, based on clear evidence of endocrine system disruption and reproductive toxicity [8; 11; 12]. These regulatory milestones reflect a broader global trend toward precautionary governance of EDCs, though enforcement and scope vary across jurisdictions.
In response, industry has shifted toward alternative plasticizers such as di(isononyl)cyclohexane-1,2-dicarboxylate (DINCH) and di(2-ethylhexyl) terephthalate (DEHTP), marketed as safer substitutes [4; 13]. Yet questions remain about whether these replacements truly eliminate health risks, or whether they represent a “regrettable substitution” in which one hazardous compound is replaced by another with similar biological effects. The ongoing debate highlights the complexity of balancing industrial utility with public health protection, and underscores the need for continuous scientific evaluation and adaptive regulatory frameworks.
Timeline of Phthalate Use and Regulation
Phthalates were first synthesized in the 1920s and adopted as plasticizers by the 1930s, becoming essential to PVC-based consumer goods. Their use expanded rapidly through the mid-20th century, embedding them in everyday products. Regulatory concern emerged in the 1990s, with Health Canada classifying DEHP as toxic in 1994. The EU followed in 1999 with restrictions on six plasticizers, and the US banned DEHP, DBP, and BBP in children’s products in 2008. By the 2010s, the EU had designated several phthalates as substances of very high concern. Industry has since shifted toward alternatives like DINCH and DEHTP, though their safety remains under scrutiny. This timeline underscores the evolving balance between industrial utility and public health protection (Figure 1).
Applications and Sources of Exposure
The industrial versatility of phthalates stems from their ability to modify the physical properties of plastics, particularly PVC, by enhancing flexibility, durability, and resistance to wear [5; 8;14]. This functional role has made them indispensable in the production of a wide range of materials, from flooring, cables, and wall coverings to medical tubing and blood bags. However, the same utility has also led to their incorporation into an astonishing array of consumer products that permeate daily life.
Phthalates are commonly found in packaging materials, food storage containers, and cling films, where they can migrate into food and beverages. They are also widely used in personal care and cosmetic products, such as shampoos, lotions, perfumes, and nail polishes, where they act as solvents, stabilisers, and fragrance carriers. Household items including detergents, adhesives, and paints frequently contain phthalates, while exposure can also occur through dust particles released from vinyl flooring, furniture, and electronic devices [2].
Because phthalates are not chemically bound to the products in which they are used, they leach readily into the environment, contaminating air, water, and soil [8; 14; 15]. This mobility creates multiple pathways of human exposure: ingestion of contaminated food and drink, inhalation of indoor dust and ambient air, and dermal absorption through direct contact with treated materials. As a result, exposure is chronic and cumulative, beginning in utero and continuing throughout the lifespan, with measurable levels detected in blood, urine, and breast milk across populations worldwide.
Classification by Molecular Weight
Phthalates are commonly classified according to molecular weight, a distinction that correlates closely with their functional applications.
Low-molecular-weight phthalates, including diethyl phthalate (DEP), dibutyl phthalate (DBP), and diisobutyl phthalate (DiBP), typically act as solvents or additives in non-PVC contexts. They are widely incorporated into cosmetics, personal care products, and fragrances, where they stabilise formulations and enhance scent longevity [2; 4; 5; 13; 14; 15]. Beyond consumer goods, these compounds also appear in certain medical devices and pharmaceuticals, such as enteric-coated tablets designed to withstand gastric acidity.
High-molecular-weight phthalates, such as di(2-ethylhexyl) phthalate (DEHP), diisononyl phthalate (DiNP), and benzyl butyl phthalate (BBP), are used primarily as plasticizers in PVC polymers. Their applications span food packaging, flooring, wall coverings, toys, and furniture, where they impart flexibility and durability [2; 4; 8; 12; 15]. Critically, they are also integral to medical equipment, including blood bags, intravenous infusion sets, and respiratory tubing, where their plasticizing properties enable safe and functional design.
Routes of Human Exposure
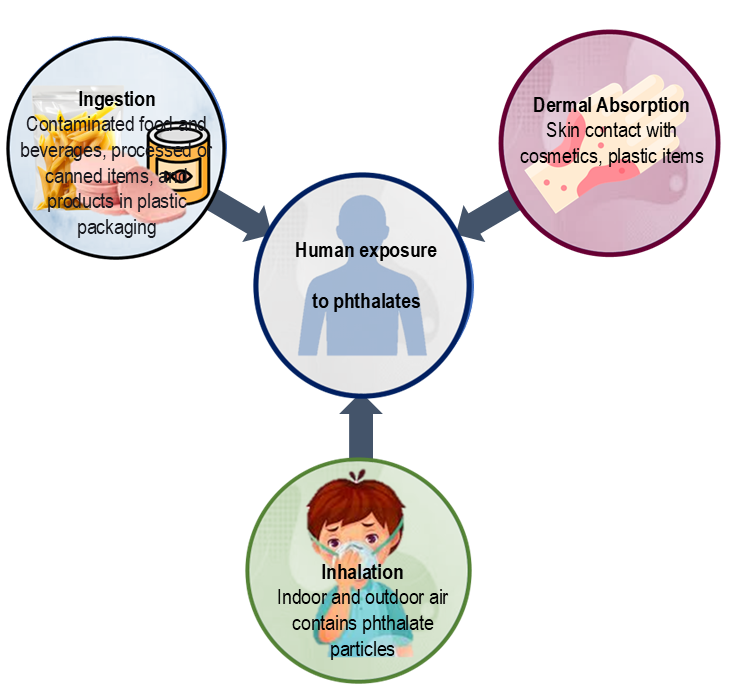
A fundamental characteristic of phthalates creates the conditions for widespread exposure; they are not chemically bound to the materials they modify. This non-covalent association allows them to leach readily into surrounding environments and into the very products they are intended to stabilise, making human exposure essentially unavoidable and chronic in nature [4; 7; 8;14; 15].
Ingestion represents the primary route of exposure for most populations. Contaminated food and beverages, particularly fatty foods, processed or canned items, and products stored in plastic packaging, serve as major sources [7; 8; 16]. The lipophilic nature of phthalates means they preferentially partition into high-fat matrices, concentrating exposure through dietary pathways. Infants and young children are especially vulnerable, as phthalates can migrate into formula, baby food, and breast milk, thereby establishing exposure early in life.
Inhalation and dermal contact provide additional, often underestimated, routes of exposure. Indoor and outdoor air contains phthalate particles classified as semi-volatile organic compounds (SVOCs), while household dust can harbour significant concentrations, particularly in environments with vinyl flooring, synthetic furniture, or electronic equipment. Direct skin contact with personal care products, cosmetics, and plastic household items further adds to the total body burden [4; 7; 8]. Occupational settings, such as manufacturing plants or healthcare environments where PVC medical devices are frequently handled, can amplify both inhalation and dermal pathways.
Taken together, these routes illustrate the multifaceted and cumulative nature of phthalate exposure. Because ingestion, inhalation, and dermal absorption occur simultaneously and continuously, phthalates contribute to a persistent internal exposure profile that spans the entire human lifespan (Figure 2).
Vulnerable Populations
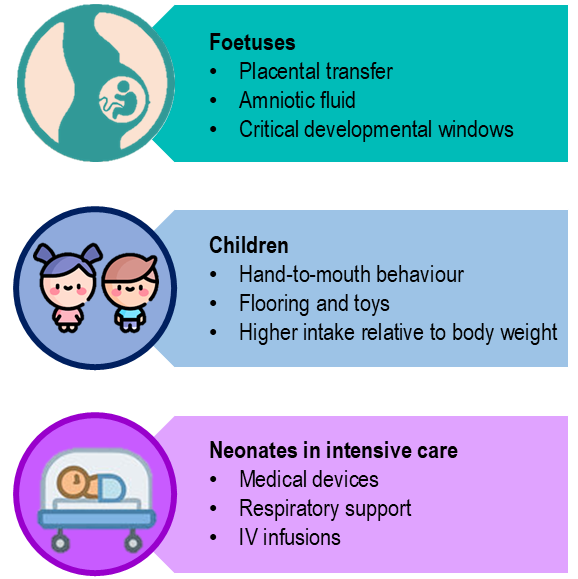
Certain groups face disproportionate exposure risks due to physiological sensitivity, developmental stage, and environmental context. Foetuses represent a particularly vulnerable population, as phthalates readily cross the placental barrier and have been detected in amniotic fluid, cord blood, and placental tissue. This creates in utero exposure during critical windows of organogenesis and neurodevelopment, when hormonal signalling plays a foundational role in shaping long-term health trajectories [2; 5; 13;17].
Infants and young children continue to face elevated risks postnatally due to their immature metabolic systems, rapid developmental pace, and behavioural patterns such as hand-to-mouth contact with plastic toys, household items, and dust-laden surfaces. Their higher surface-area-to-body-weight ratio and increased dietary intake per kilogram further amplify exposure [5; 13]. Moreover, children’s environments, daycare centres, schools, and homes, often contain phthalate-rich materials such as vinyl flooring, synthetic furniture, and fragranced products.
Neonates in intensive care settings encounter a unique and substantial exposure scenario. Those admitted to Neonatal Intensive Care Units (NICUs) may receive considerable doses of phthalates through essential medical devices, including intravenous tubing, catheters, respiratory masks, and blood bags. These devices, often made from PVC softened with DEHP, leach phthalates directly into the bloodstream during critical periods of vulnerability. This creates a paradox wherein life-saving interventions simultaneously expose the most fragile patients to endocrine-disrupting chemicals [12].
Collectively, these populations underscore the ethical and clinical imperative to reduce phthalate exposure in settings where vulnerability is heightened and alternatives are increasingly available (Figure 3).
Metabolism and Biological Fate
Once absorbed, phthalates undergo relatively rapid metabolism, with biological half-lives typically under 24 hours [2; 5; 15]. In the liver and other tissues, they are quickly hydrolysed to monoesters, metabolites that often exhibit greater biological activity than their parent compounds. These monoesters can bind to hormone receptors and disrupt endocrine signalling, making them central to the toxicological profile of phthalates. Following initial hydrolysis, the monoesters undergo further oxidative transformation and conjugation (e.g., glucuronidation), facilitating their excretion primarily via urine [5; 7; 14].
While this rapid metabolic turnover might suggest limited opportunity for harm, the reality is more complex. The chronic and repeated nature of exposure, through diet, air, dust, and skin contact, means that the body is continuously re-exposed before it can fully eliminate previous doses [14]. This creates a pseudo-steady-state condition in which biologically active metabolites are present in tissues and fluids at all times. Biomonitoring studies consistently detect phthalate metabolites in urine samples across all age groups [5; 7; 14], including pregnant. women and neonates, underscoring the persistence of exposure despite short half-lives. The disconnect between rapid clearance and sustained internal burden highlights the need to evaluate phthalate toxicity not just by pharmacokinetics, but by cumulative exposure and vulnerable timing.
Mechanisms of Endocrine Disruption
Phthalates exert their effects as endocrine disruptors through multiple, often overlapping mechanisms, acting as agonists, antagonists, or modulators across diverse hormonal signalling pathways. Their structural similarity to endogenous hormones allows them to bind to nuclear receptors and interfere with transcriptional regulation, leading to altered gene expression and disrupted feedback loops.
Key molecular targets include steroid hormone receptors, such as oestrogen receptors (ER) and androgen receptors (AR), where phthalates can mimic or block natural ligands, leading to imbalances in reproductive hormone signalling. This interference has been linked to altered sexual development, reduced fertility, and changes in secondary sex characteristics [6; 18].
Phthalates also interact with thyroid hormone receptors (ThR), disrupting the hypothalamic-pituitary-thyroid axis and affecting metabolic regulation, neurodevelopment, and thermogenesis [19]. These disruptions are particularly concerning during gestation and early childhood, when thyroid hormones are critical for brain maturation and growth.
Another important class of targets includes peroxisome proliferator-activated receptors (PPARs), which regulate lipid metabolism, adipogenesis, and insulin sensitivity. Phthalate-induced modulation of PPARs has been implicated in metabolic disorders such as obesity, insulin resistance, and type 2 diabetes [1].
This multi-receptor interference contributes to widespread hormone dysregulation, affecting reproductive, metabolic, neurological, and immune systems. The pleiotropic nature of phthalate action, combined with chronic exposure, amplifies their potential to disrupt endocrine homeostasis across the lifespan.
Health Impacts Across the Lifespan
The health consequences of phthalate exposure manifest across multiple organ systems and life stages, creating a pattern of dysfunction that can originate before birth and persist throughout life.
Reproductive and Developmental Effects
Male reproductive health emerges as particularly sensitive to phthalate exposure. Compounds such as DEHP and DBP act as anti-androgens [3; 13], reducing testosterone levels during critical developmental periods. This hormonal disruption can lead to a constellation of abnormalities including reduced anogenital distance, cryptorchidism (undescended testicles), and hypospadias (urethral opening abnormalities), collectively termed "phthalate syndrome" in animal models [6; 13; 16]. Beyond developmental impacts, adult male fertility suffers through reduced sperm quality, decreased motility and concentration, and increased DNA fragmentation in sperm cells [20].
Female reproductive health shows its own pattern of vulnerability. Phthalate exposure associates significantly with endometriosis, a painful condition characterised by chronic inflammation and abnormal growth of endometrial tissue outside the uterus. Specific phthalate metabolites including MEHP (mono-(2-ethylhexyl) phthalate) and MBzP (monobenzyl phthalate) show particular associations with this condition [3; 7; 21]. Additionally, phthalates can disrupt the process of follicle development in ovaries, potentially contributing to diminished ovarian reserve and negatively affecting outcomes in assisted reproduction procedures such as in vitro fertilization [14].
Pregnancy outcomes represent another area of concern. Maternal phthalate exposure correlates with increased risk of preterm birth, with metabolites including MEP (monoethyl phthalate), MECPP (mono-(2-ethyl-5-carboxypentyl) phthalate), MBzP, and DEHP showing particular associations [4; 22]. Exposure also links to pregnancy loss and low birth weight [13; 14], outcomes that carry lifelong consequences for affected children.
Metabolic and Endocrine Disorders
The metabolic impacts of phthalates extend far beyond reproductive effects. These chemicals function as "obesogens," interfering with the regulation of body mass and promoting adipogenesis, the formation of fat tissue [6; 8;14]. Population studies demonstrate associations between phthalate exposure and increased body mass index, greater waist circumference, and metabolic syndrome [6; 8; 23].
Diabetes risk increases with phthalate exposure through mechanisms involving insulin resistance. The association with Type 2 Diabetes Mellitus appears to operate through multiple pathways, including activation of PPARs that regulate lipid and glucose metabolism, as well as induction of oxidative stress that damages insulin-producing cells and insulin-responsive tissues [2; 24].
Thyroid function also suffers disruption. Multiple phthalate metabolites including MECPP, MEHHP (mono-(2-ethyl-5-hydroxyhexyl) phthalate), MEHP, and MEOHP (mono-(2-ethyl-5-oxohexyl) phthalate) show significant associations with decreased serum thyroxine levels in adults, indicating interference with thyroid hormone production or metabolism [19].
Carcinogenic Potential and Other Toxicological Concerns
The Environmental Protection Agency classifies DEHP as a probable human carcinogen, while the International Agency for Research on Cancer considers it possibly carcinogenic to humans [11]. Epidemiological evidence supports these classifications, with exposure to DBP and DEHP showing significant associations with increased risk of invasive breast cancer [15; 25].
Beyond direct carcinogenic effects, phthalates contribute to the broader toxic burden of air pollution. Particulate DEHP in ambient air represents a significant contributor to the endocrine-disrupting potential of fine particulate matter (PM₂.₅) [26], adding to the health risks of air pollution beyond its well-established cardiovascular and respiratory effects.
Neurodevelopmental impacts in children add another dimension of concern. Studies link phthalate exposure to poorer performance on cognitive assessments, including deficits in visual reception and fine motor skills. MEP and certain DEHTP metabolites during infancy show particular associations with these outcomes [13; 27], raising concerns about long-term impacts on learning and development.
A Systems Perspective on Phthalate Toxicity
The extensive and interconnected pathways affected by phthalate exposure create a pattern of systemic dysfunction that transcends isolated organ effects. Rather than targeting a single receptor or tissue, phthalates interfere with hormonal signalling networks that regulate development, metabolism, reproduction, and neuroendocrine function. Continuous exposure acts analogously to introducing a wrench into delicate hormonal machinery, disrupting feedback loops, receptor sensitivity, and gene expression across multiple systems. This disruption propagates through endocrine axes such as the hypothalamic-pituitary-gonadal (HPG), hypothalamic-pituitary-thyroid (HPT), and hypothalamic-pituitary-adrenal (HPA) systems, creating cascading effects that can persist and manifest across the entire lifespan [14].
The consequences of such interference are often amplified during critical windows of vulnerability, gestation, infancy, puberty, when hormonal cues orchestrate tissue differentiation, brain development, and metabolic programming. Even low-level exposure during these periods can reprogram physiological set points, increasing susceptibility to chronic conditions such as infertility, obesity, diabetes, and neurodevelopmental disorders later in life.
This situation mirrors how chronic, low-level pollution in a vital river system can destabilize the health of an entire ecosystem. Just as contaminants introduced upstream can accumulate, interact, and affect species far downstream, phthalates introduced into the human body, through diet, air, or skin, can ripple through biological systems, altering trajectories long after the initial exposure. The systemic nature of phthalate toxicity demands a holistic approach to risk assessment, one that accounts for cumulative exposure, developmental timing, and cross-system interactions rather than isolated endpoints.
Conclusion
Phthalates represent a complex challenge at the intersection of industrial utility and public health. The very chemical properties that make them indispensable in manufacturing, flexibility, durability, and ease of incorporation into diverse products, are also the traits that enable their continual release into the environment and, ultimately, into the human body. Despite decades of evolving regulation and mounting scientific evidence, the fundamental problem of ubiquitous exposure remains unresolved.
To understand phthalates is to recognise that they are not simply isolated compounds with discrete effects, but a class of chemicals capable of disrupting hormonal signalling across multiple physiological systems. Their impacts begin before birth, crossing the placental barrier, and extend throughout the lifespan, influencing reproduction, metabolism, neurodevelopment, and even cancer risk. This systemic reach underscores their significance as more than just industrial additives; they are agents of biological disruption woven into the fabric of daily life.
As science continues to clarify the breadth of phthalate toxicity and as alternative plasticizers enter the marketplace, the critical question persists: how can society reconcile the undeniable utility of these chemicals with their demonstrated capacity to undermine human health at its most fundamental, hormonal level?
References
- Li, Y., Yang, H., He, W., & Li, Y. (2023). Human endocrine-disrupting effects of phthalate esters through adverse outcome pathways: A comprehensive mechanism analysis. International Journal of Molecular Sciences, 24(17), 13548. https://doi.org/10.3390/ijms241713548
- Mariana, M., & Cairrao, E. (2023). The relationship between phthalates and diabetes: A review. Metabolites, 13(6), 746. https://doi.org/10.3390/metabo13060746
- Interdonato, L., Siracusa, R., Fusco, R., Cuzzocrea, S., & Di Paola, R. (2023). Endocrine disruptor compounds in environment: Focus on women’s reproductive health and endometriosis. International Journal of Molecular Sciences, 24(6), 5682. https://doi.org/10.3390/ijms24065682
- Martin, L., Zhang, Y., First, O., Mustieles, V., Dodson, R., Rosa, G., Coburn-Sanderson, A., Adams, C. D., & Messerlian, C. (2022). Lifestyle interventions to reduce endocrine-disrupting phthalate and phenol exposures among reproductive age men and women: A review and future steps. Environment International, 170, 107576. https://doi.org/10.1016/j.envint.2022.107576
- Wang, Y., & Qian, H. (2021). Phthalates and their impacts on human health. Healthcare, 9(5), 603. https://doi.org/10.3390/healthcare9050603
- Predieri, B., Iughetti, L., Bernasconi, S., & Street, M. E. (2022). Endocrine disrupting chemicals’ effects in children: What we know and what we need to learn. International Journal of Molecular Sciences, 23(19), 11899. https://doi.org/10.3390/ijms231911899
- Ribeiro, B., Mariana, M., Lorigo, M., Oliani, D., Ramalhinho, A. C., & Cairrao, E. (2024). Association between the exposure to phthalates and the risk of endometriosis: An updated review. Biomedicines, 12(8), 1932. https://doi.org/10.3390/biomedicines12081932
- Dalamaga, M., Kounatidis, D., Tsilingiris, D., Vallianou, N. G., Karampela, I., Psallida, S., & Papavassiliou, A. G. (2024). The role of endocrine disruptors bisphenols and phthalates in obesity: Current evidence, perspectives and controversies. International Journal of Molecular Sciences, 25, 675. https://doi.org/10.3390/ijms25010675
- Urmi, M. A., Akbor, M. A., Sarker, S., Nahar, A., Shaikh, M. A. A., Siddique, M. A. B., Ahmed, S., Meghna, A. J., Malafaia, G., & Rahman, M. M. (2023). A pioneering study on endocrine disruptors (phthalates esters) in urban rivers of Bangladesh: An appraisal of possible risk assessment to ecology and human health. Journal of Hazardous Materials Advances, 12, 100369. https://doi.org/10.1016/j.hazadv.2023.100369
- Li, Z., & Robaire, B. (2025). Effects of endocrine-disrupting chemicals on adrenal function. Endocrinology, 166, bqaf045. https://doi.org/10.1210/endocr/bqaf045
- Alsen, M., Sinclair, C., Cooke, P., Ziadkhanpour, K., Genden, E., & van Gerwen, M. (2021). Endocrine disrupting chemicals and thyroid cancer: An overview. Toxics, 9(1), 14. https://doi.org/10.3390/toxics9010014
- Panneel, L., Cleys, P., Poma, G., Ait Bamai, Y., Jorens, P. G., Covaci, A., & Mulder, A. (2024). Ongoing exposure to endocrine disrupting phthalates and alternative plasticizers in neonatal intensive care unit patients. Environment International, 186, 108605. https://doi.org/10.1016/j.envint.2024.108605
- Lucaccioni, L., Trevisani, V., Passini, E., Righi, B., Plessi, C., Predieri, B., & Iughetti, L. (2021). Perinatal exposure to phthalates: From endocrine to neurodevelopment effects. International Journal of Molecular Sciences, 22(8), 4063. https://doi.org/10.3390/ijms22084063
- Basso, C. G., de Araújo-Ramos, A. T., & Martino-Andrade, A. J. (2022). Exposure to phthalates and female reproductive health: A literature review. Reproductive Toxicology, 109, 61–79. https://doi.org/10.1016/j.reprotox.2022.02.006
- Benoit, L., Tomkiewicz, C., Bortoli, S., Bats, A. S., Coumoul, X., & Koual, M. (2025). Role of phthalates in breast cancer initiation, progression and drug resistance: A scoping review and recommendations. Toxicology Letters, 413, 111721. https://doi.org/10.1016/j.toxlet.2025.111721
- Sharpe, R. M. (2024). Endocrine disruption and male reproductive disorders: Unanswered questions. Human Reproduction, 39(9), 1879–1888. https://doi.org/10.1093/humrep/deae143
- Bräuner, E. V., Uldbjerg, C. S., Lim, Y.-H., Gregersen, L. S., Krause, M., Frederiksen, H., & Andersson, A.-M. (2022). Presence of parabens, phenols and phthalates in paired maternal serum, urine and amniotic fluid. Environment International, 158, 106987. https://doi.org/10.1016/j.envint.2021.106987
- Martínez-Pinna, J., Sempere-Navarro, R., Medina-Gali, R. M., Fuentes, E., Quesada, I., Sargis, R. M., Trasande, L., & Nadal, A. (2023). Endocrine disruptors in plastics alter β-cell physiology and increase the risk of diabetes mellitus. American Journal of Physiology-Endocrinology and Metabolism, 324(4), E488–E505. https://doi.org/10.1152/ajpendo.00068.2023
- Gao, Q., Song, Y., Jia, Z., Huan, C., Cao, Q., Wang, C., Mao, Z., & Huo, W. (2024). Association of exposure to a mixture of phenols, parabens, and phthalates with altered serum thyroid hormone levels and the roles of iodine status and thyroid autoantibody status: A study among American adults. Ecotoxicology and Environmental Safety, 282, 116754. https://doi.org/10.1016/j.ecoenv.2024.116754
- Virant-Klun, I., Imamovic-Kumalic, S., & Pinter, B. (2022). From oxidative stress to male infertility: Review of the associations of endocrine-disrupting chemicals (bisphenols, phthalates, and parabens) with human semen quality. Antioxidants, 11, 1617. https://doi.org/10.3390/antiox11081617
- Chitakwa, N., Alqudaimi, M., Sultan, M., & Wu, D. (2024). Plastic-related endocrine disrupting chemicals significantly related to the increased risk of estrogen-dependent diseases in women. Environmental Research, 252, 118966. https://doi.org/10.1016/j.envres.2024.118966
- Wu, Y., Wang, J., Wei, Y., Chen, J., Kang, L., Long, C., Wu, S., Shen, L., & Wei, G. (2022). Maternal exposure to endocrine disrupting chemicals (EDCs) and preterm birth: A systematic review, meta-analysis, and meta-regression analysis. Environmental Pollution, 292, 118264. https://doi.org/10.1016/j.envpol.2021.118264
- Biemann, R., Blüher, M., & Isermann, B. (2021). Exposure to endocrine-disrupting compounds such as phthalates and bisphenol A is associated with an increased risk for obesity. Best Practice & Research Clinical Endocrinology & Metabolism, 35, 101546. https://doi.org/10.1016/j.beem.2021.101546
- Hinault, C., Caroli-Bosc, P., Bost, F., & Chevalier, N. (2023). Critical overview on endocrine disruptors in diabetes mellitus. International Journal of Molecular Sciences, 24(5), 4537. https://doi.org/10.3390/ijms24054537
- Mukherjee Das, A., Gogia, A., Garg, M., Elaiyaraja, A., Arambam, P., Mathur, S., Babu-Rajendran, R., Deo, S. V. S., Kumar, L., Das, B. C., & Janardhanan, R. (2022). Urinary concentration of endocrine-disrupting phthalates and breast cancer risk in Indian women: A case-control study with a focus on mutations in phthalate-responsive genes. Cancer Epidemiology, 79, 102188. https://doi.org/10.1016/j.canep.2022.102188
- Zhou, Q., Chen, J., Zhang, J., Zhou, F., Zhao, J., Wei, X., Zheng, K., Wu, J., Li, B., & Pan, B. (2022). Toxicity and endocrine-disrupting potential of PM2.5: Association with particulate polycyclic aromatic hydrocarbons, phthalate esters, and heavy metals. Environmental Pollution, 292, 118349. https://doi.org/10.1016/j.envpol.2021.118349
- Sotelo-Orozco, J., Calafat, A. M., Botelho, J. C., Schmidt, R. J., Hertz-Picciotto, I., & Bennett, D. H. (2024). Exposure to endocrine disrupting chemicals including phthalates, phenols, and parabens in infancy: Associations with neurodevelopmental outcomes in the MARBLES study. International Journal of Hygiene and Environmental Health, 261, 114425. https://doi.org/10.1016/j.ijheh.2024.114425
