PFAS: The Chemistry Behind the “Forever Chemicals”

What Are PFAS?
Per- and polyfluoroalkyl substances (PFAS) are a large family of man-made chemicals first developed in the late 1930s and widely used from the 1940s onward. Nicknamed “forever chemicals” because they do not break down easily in nature or in our bodies, PFAS persist for decades once produced. Their unique ability to resist heat, water, and oil led to their use in everyday products such as non-stick cookware, food packaging, water-resistant clothing, and firefighting foams. Over time, this persistence has resulted in widespread contamination, raising growing concerns about long-term health effects, including potential disruption of hormonal systems.
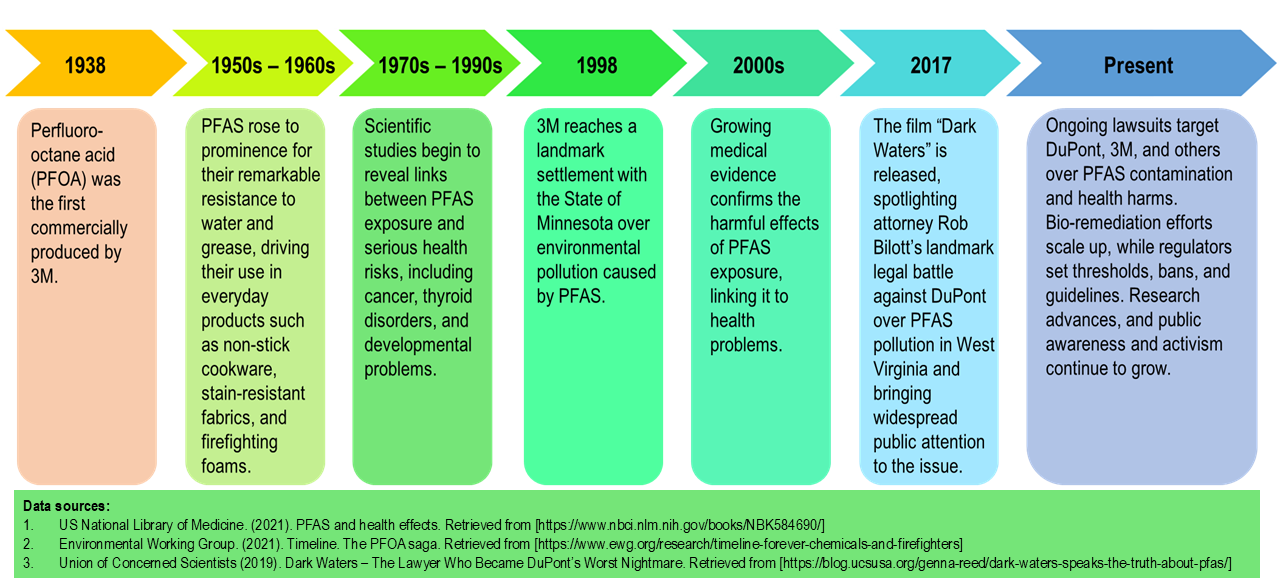
Timeline of PFAS development from 1938 till present (adapted from Lee et al. 2025)
PFAS Acronyms Explained
Acronym | Full Name | Notes |
PFAS | Per- and polyfluoroalkyl substances | Large family of synthetic chemicals, often called “forever chemicals.” |
PFOA | Perfluorooctanoic acid | Once widely used in Teflon™ and stain-resistant products; linked to cancer and immune disorders. |
PFOS | Perfluorooctanesulfonic acid | Common in firefighting foams, stain-resistant fabrics, and food packaging; highly persistent and toxic. |
PFAS “forever” Secret Lies in Their Structure
PFAS are a group of synthetic organofluorine chemical compounds comprising of a tail and a head. PFAS molecules share a unique design:
- Hydrophobic (water repelling) Tail: A carbon chain where hydrogen atoms are replaced by fluorine (perfluorinated), making the molecules incredibly stable and resistant to heat, water, and oil.
- Hydrophilic Head: The tail is linked to a water-loving (hydrophilic) functional group (head) such as carboxylic acid or sulfonic acid, creating "forever chemicals" known for stability and widespread use in non-stick, waterproof, and firefighting products.
This dual nature makes PFAS amphiphilic, which means they can repel both water and oil. That is why they are used in non‑stick pans, waterproof jackets, and grease‑resistant packaging.
PFAS chemicals can be straight chains (such as PFOA, PFOS) or branched, leading to over 12,000 known PFAS compounds.
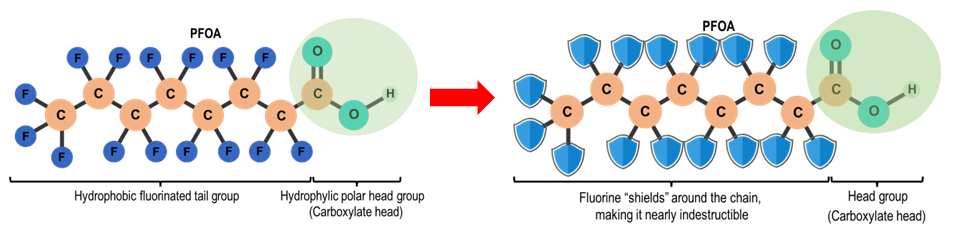
Chemical structure of PFAS (PFOA)
Why “Forever”?
The strength of PFAS lies in the carbon–fluorine (C–F) bond:
- It is the strongest single bond in organic chemistry.
- It is short, tight, and extremely hard to break.
- It is resistant to heat, acids, bases, and biological attack.
Types and Classifications of PFAS
PFAS are not all the same. Scientists group them into categories based on their chemical structure and behaviour. Understanding these differences helps explain why some PFAS are more dangerous than others, and why they show up in different products.
- Perfluoroalkyl vs. polyfluoroalkyl
- Perfluoroalkyl: fully fluorinated carbon chains.
- Polyfluoroalkyl: partially fluorinated chains.
- Chain length
- Long-chain PFAS (e.g., PFOA, PFOS): tend to build up in the body and environment, making them more bioaccumulative.
- Short-chain PFAS (e.g., PFBS, PFBA): move more easily through water and soil, so they spread faster, but they are still toxic.
- Polymers vs. non-polymers
- Non-polymeric PFAS (e.g., PFAAs): highly mobile and persistent, often detected in drinking water and human blood.
- Polymeric PFAS (e.g., Teflon/PTFE): used in cookware, coatings, and textiles; less mobile but still resistant to breakdown.
PFAS types and their characteristics
Classification | Examples | Key Features | Common Uses | Risks |
Perfluoroalkyl | PFOA, PFOS | Fully fluorinated chains | Non-stick cookware, firefighting foams | Highly persistent, bioaccumulative |
Polyfluoroalkyl | PFBS, PFBA | Partially fluorinated chains | Food packaging, textiles | More mobile in water, still toxic |
Long-chain PFAS | PFOA, PFOS | Longer carbon chains | Industrial coatings, stain-resistant fabrics | Build up in humans and wildlife |
Short-chain PFAS | PFBS, PFBA | Shorter carbon chains | Packaging, water-repellent clothing | Spread quickly, toxic but less bioaccumulative |
Non-polymeric PFAS | PFAAs | Mobile, persistent | Detected in water and blood | Long-term contamination |
Polymeric PFAS | Teflon/PTFE | Large molecules, less mobile | Cookware, coatings, textiles | Resistant to breakdown, environmental persistence |
Precursors and Transformation
Not all PFAS are stable from the start. Some act as precursors - chemicals that slowly break down over time into more permanent PFAS forms. These precursors can transform into terminal compounds such as PFOA and PFOS, which are even more resistant to degradation.
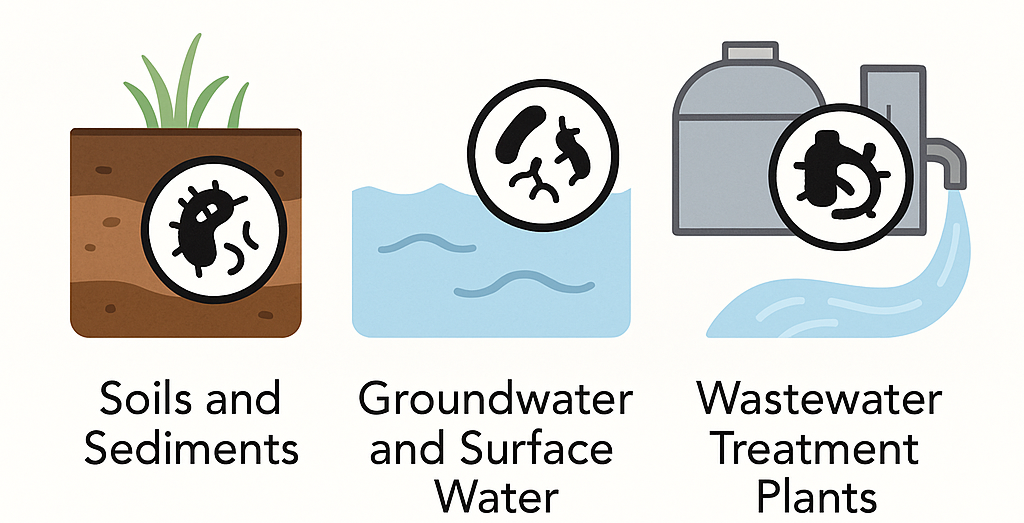
Microbial biotransformation of PFAS precursors takes place in natural environments where microbes are active, especially in:
- Soils and sediments
- Groundwater and surface water
- Wastewater treatment plants
Main Transformation Pathways:
Microbial biotransformation: Certain microbes can slowly break down precursor PFAS, step by step, into simpler and more stable perfluoroalkyl acids. This process is usually incomplete, but it steadily produces persistent PFAS that resist further degradation.
Chemical transformation: In soil and water, precursors can undergo oxidation or hydrolysis reactions that strip away parts of their structure, leaving behind highly stable PFAS end-products.
Sorption and release: Precursors may attach (sorb) to soil or sediment surfaces, where they can later desorb and continue transforming. This slows their movement but prolongs contamination.
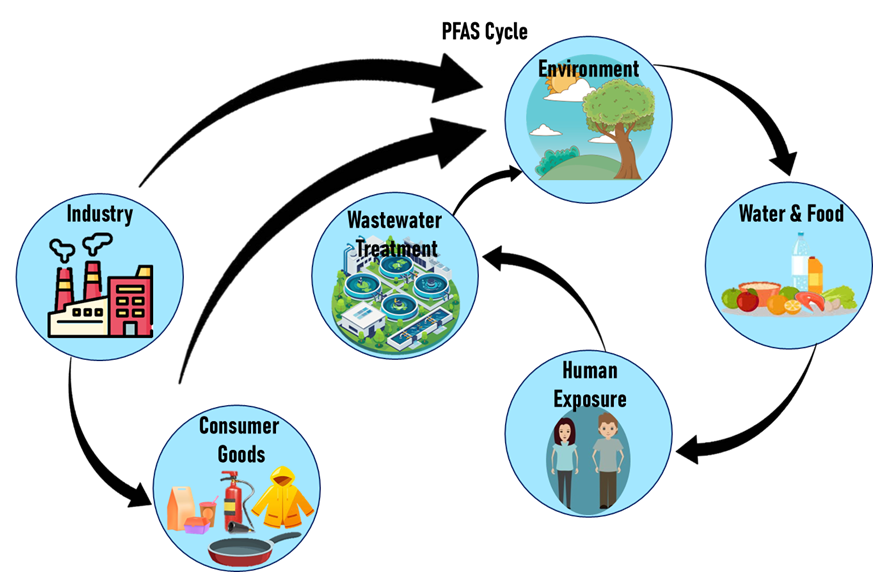
PFAS precursors transform in soil, water, and treatment systems into persistent PFAS
Why This Matters
Understanding the chemistry of PFAS explains why they are so persistent and why regulation is difficult. Their structure makes them useful in industry but nearly impossible to remove once released. The strong carbon-fluorine bonds that give PFAS their durability also make them resistant to heat, water, and chemical breakdown - properties that are prized in manufacturing but problematic in nature. Once PFAS enter the environment, they do not degrade easily, accumulate over time, and can travel through soil and water. This persistence means that even small releases can lead to long-term contamination, making cleanup costly and complex.
Bibliography
Lee, J.C.; Smaoui, S.; Duffill, J.; Marandi, B.; Varzakas, T. Research Progress in Current and Emerging Issues of PFASs’Global Impact: Long-Term Health Effects and Governance of Food Systems. Foods 2025, 14, 958. https://doi.org/10.3390/foods14060958
