The Hidden Chemistry in Your Kitchen: What You Need to Know About Bisphenols
A Chemical That's Older Than You Think
In 1891, a Russian chemist named Alexander Dianin mixed acetone with phenol in his laboratory and created something new: bisphenol A (BPA). He had no idea that his creation would eventually become one of the most widespread synthetic chemicals on Earth, touching nearly every aspect of modern life, including yours.
Fast forward to today, and BPA (as it is commonly known) is everywhere. That reusable water bottle in your bag? Probably contains it. The lining inside your canned tomatoes? Almost certainly. Even that innocent-looking receipt from this morning's coffee run is coated with it. We are producing over 7 million tons of this stuff annually, and despite decades of health concerns, that number keeps climbing.
But here is what makes this story particularly troubling: scientists discovered BPA's ability to mimic human hormones back in the 1930s, decades before it became a staple of consumer products. We knew it could interfere with our bodies' natural signalling systems, yet we built an entire industrial ecosystem around it anyway.
The "Miracle Material" That Wasn't
BPA's rise to dominance happened for good reasons. In the 1950s and 60s, researchers discovered they could transform it into polycarbonate (PC) plastics, materials that were strong, clear, and heat-resistant. Unlike glass, they would not shatter. Unlike older plastics, they could withstand high temperatures. They seemed perfect.
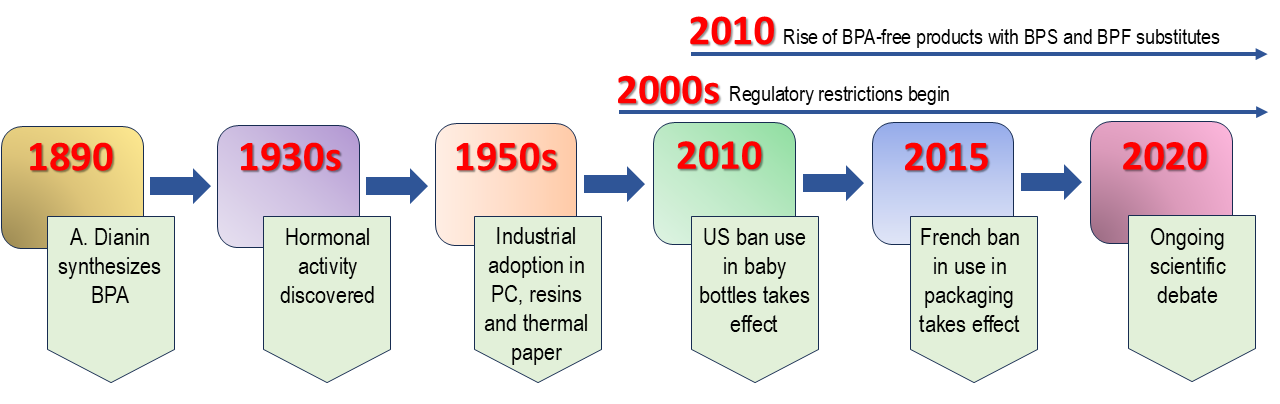
The chemical industry embraced BPA with enthusiasm. It became the foundation for:
- Polycarbonate plastics (70% of all BPA production): Water bottles, food containers, baby bottles, electronics, medical devices, and countless other products promised convenience and durability.
- Epoxy resins: These formed protective linings inside metal cans, keeping your food from tasting metallic and preventing corrosion. They sealed water pipes and even became part of dental fillings.
- Thermal paper: That is the coating on receipts that makes them print without ink, and it contains BPA in its free, unbound form, ready to transfer directly onto your skin.
By the late 20th century, BPA had become synonymous with modern convenience. It was hailed as a miracle of chemistry. But miracles, as we have learned, often come with unintended consequences.
The Problem: Your Body Thinks BPA Is a Hormone
Here is where the science gets concerning. BPA's molecular structure closely resembles oestrogen, specifically, the natural hormone 17β-estradiol that regulates countless functions in your body. This is not a coincidence; it's chemistry. The two hydroxyl groups on BPA sit in just the right positions to fool your body's oestrogen receptors.
When BPA enters your system (and it does, regularly, through food, water, and skin contact), it can bind to these receptors and trigger responses your body never intended. Sometimes it acts like oestrogen, switching on biological processes. Other times it blocks real oestrogen from doing its job. This interference with your endocrine system, your body's chemical messaging network, is why BPA is classified as an endocrine-disrupting chemical (EDC).
The effects ripple outward: metabolic disruption, reproductive issues, developmental problems in children, and links to conditions from obesity to diabetes. And because BPA loves fat (it is highly lipophilic), it does not just pass through your system, it can accumulate in your tissues.
The Great BPA Switcheroo
As evidence mounted and parents grew alarmed about baby bottles and Sippy cups, regulations began restricting BPA use in certain products. The chemical industry's response? Substitution. Enter BPS (bisphenol S) and BPF (bisphenol F), structurally similar cousins that allowed manufacturers to slap "BPA-free" labels on their products.
Problem solved, right?
Not even close. Scientists call this "regrettable substitution," and it is exactly what it sounds like. BPS and BPF turn out to have similar, and sometimes worse, endocrine-disrupting effects than BPA itself. We essentially traded one problematic chemical for two others with the same issues, then congratulated ourselves with reassuring labels.
Those BPA-free receipts you have been accepting without worry? Many now contain BPS instead, which still transfers onto your skin and into your body. That BPA-free water bottle? It might contain BPF, which some studies suggest has even greater obesogenic potential than the original.
The "BPA-free" label has become a masterclass in false reassurance, technically accurate but practically meaningless when the replacements pose comparable risks.
Where Are You Being Exposed?
The honest answer is: almost everywhere. But some sources are particularly significant:
- Your kitchen is ground zero. Canned foods and beverages are major culprits, as BPA leaches from the epoxy linings, especially when contents are acidic (like tomatoes) or when cans are heated. Polycarbonate containers release more BPA when microwaved or when they are scratched and aging. Even cow's milk and dairy products show contamination.
- Your wallet is another problem. Every time you handle a receipt, free BPA (or BPS) transfers to your fingers. Use hand sanitiser or lotion afterward? That increases absorption through your skin. Cashiers and retail workers face occupational exposure from handling hundreds of receipts daily.
- Your home environment accumulates bisphenols in dust, released from electronics, plastics, and thermal paper. Children playing on floors face higher exposure through hand-to-mouth contact.
- Your water may be contaminated through industrial runoff, landfill leachate, and even the pipes carrying it to your tap. Bottled water is not necessarily safer, those plastic bottles can leach bisphenols too.
The result is what scientists call "ubiquitous exposure." You are not encountering BPA occasionally; you are encountering it constantly, through multiple pathways, often without realizing it.
Why This Matters Now
We are living with the consequences of decisions made decades ago when the long-term health implications of widespread chemical exposure were not well understood, or were not prioritised over industrial convenience. BPA's story reveals how chemicals can become so deeply embedded in manufacturing and daily life that removing them becomes extraordinarily difficult, even after we recognise their harms.
The current situation presents a dilemma: BPA and its analogues remain "indispensable" to industry because they work so well for their intended purposes. Replacing them requires finding alternatives that match their performance characteristics while actually being safer, a challenge that has largely failed so far.
Meanwhile, production continues at massive scale. At over 7 million tons annually, bisphenol exposure is not decreasing; it is expanding into new markets and applications. The chemicals that entered our supply chains in the mid-20th century are now in our bodies, our water systems, and our soil.
The Path Forward
This is not a story designed to make you throw out everything plastic in your home or avoid touching receipts forever (though declining printed receipts when you do not need them is not a bad idea). It is about understanding that the chemicals surrounding us in modern life have biological effects we are only beginning to fully comprehend.
The history of bisphenols teaches us that true solutions require more than substitution. We need chemicals designed with safety in mind from the beginning, not tested for safety after they have already saturated our environment. We need regulations that look at entire chemical classes, not just individual compounds that can be easily swapped for similar alternatives. And we need transparency about what is in the products we use daily.
More than a century after Dianin's laboratory synthesis, bisphenols remain a defining example of how industrial chemistry can quietly reshape human health and the environment. Understanding their story helps us ask better questions about the other synthetic chemicals entering our lives today, and whether we're repeating the same patterns that led us here.
The plastic world we've built is convenient, durable, and in many ways, remarkable. But it is also teaching us that some conveniences come with invisible costs that we're only beginning to count.
Want to reduce your exposure?
Start with small changes: choose fresh or frozen foods over canned, when possible, avoid microwaving plastic containers, decline receipts you do not need, and use glass or stainless-steel containers for food storage.
These steps will not eliminate exposure entirely, bisphenols are too widespread for that, but they can meaningfully reduce your daily contact with these endocrine disruptors.
Scavenger Hunt: Bisphenols in Your Kitchen
Let us turn awareness into action. This activity invites you to explore your own kitchen and uncover potential sources of bisphenol exposure. It is simple, diagnostic, and surprisingly eye-opening.

Step 1: Scan Your Space
Walk into your kitchen and take a slow look around. Focus on items made of:
- Plastic
- Resin
- Lined metal (especially cans and jar lids)
Grab a notepad or your phone and jot down every item that might fall into these categories.
Your Visual Reference: What to Look For
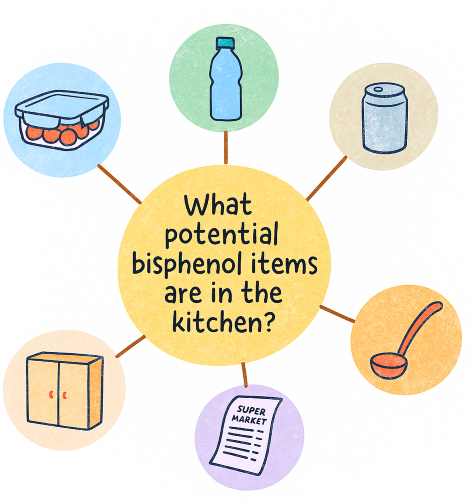
Step 2: Categorise Your Findings
Group your items into the following categories:
- Food Storage: plastic containers, cling wrap, water bottles
- Cooking Tools: spatulas, mixing bowls, cutting boards
- Packaging: canned foods, jar lids, plastic wraps
- Appliances: coffee makers, kettles with plastic interiors
Bonus: Check for recycling codes #3, #6, or #7, these often indicate bisphenol-containing plastics.
Step 3: Reflect and Act
Ask yourself:
- Which of these items do I use daily?
- Are any scratched, heated often, or aging?
- What safer alternatives could I consider?
